QUESTION IMAGE
Question
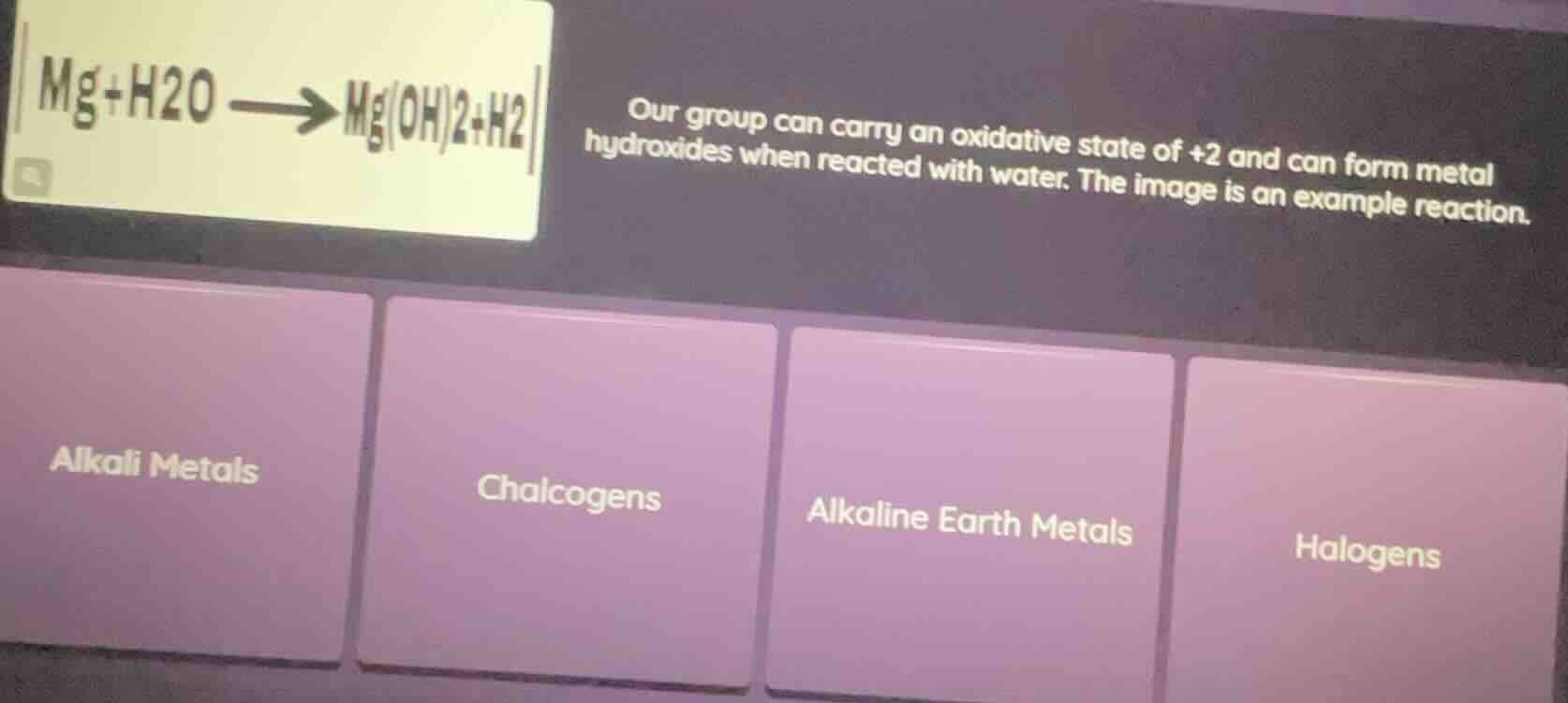
mg+h2o → mg(oh)2+h2 our group can carry an oxidative state of +2 and can form metal hydroxides when reacted with water. the image is an example reaction. alkali metals chalcogens alkaline earth metals halogens
Brief Explanations
Magnesium (Mg) has a +2 oxidation state, forms a hydroxide ($\text{Mg(OH)}_2$) with water, and is a member of the group described. This matches the defining traits of the Alkaline Earth Metals group.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Alkaline Earth Metals