QUESTION IMAGE
Question
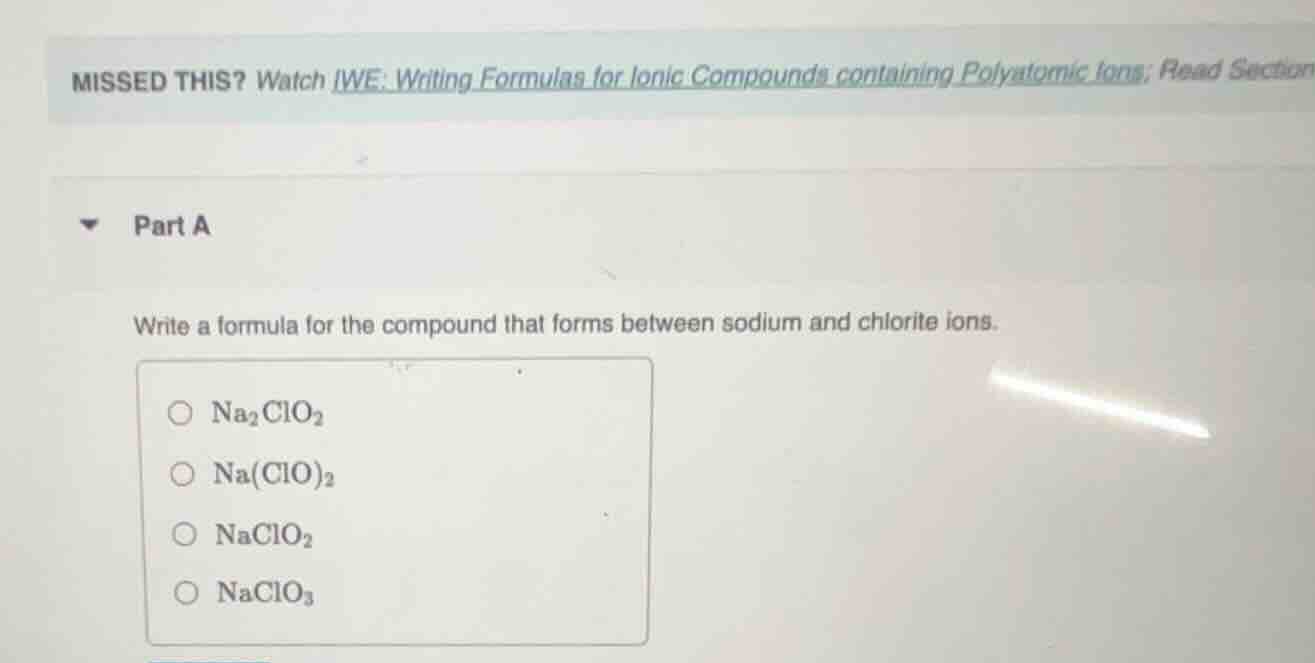
missed this? watch iwe: writing formulas for ionic compounds containing polyatomic ions; read section
part a
write a formula for the compound that forms between sodium and chlorite ions.
na₂clo₂
na(clo)₂
naclo₂
naclo₃
Step1: Determine ion charges
Sodium ion is $Na^+$ (charge +1). Chlorite ion is $ClO_2^-$ (charge -1).
Step2: Balance charges
To form a neutral compound, the number of positive and negative charges must be equal. Since $Na^+$ has +1 and $ClO_2^-$ has -1, we need 1 $Na^+$ and 1 $ClO_2^-$ to balance (1×(+1) + 1×(-1) = 0).
Step3: Write the formula
Combine the ions: $Na^+$ and $ClO_2^-$ form $NaClO_2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{NaClO_2}$ (corresponding to the option "NaClO₂")