QUESTION IMAGE
Question
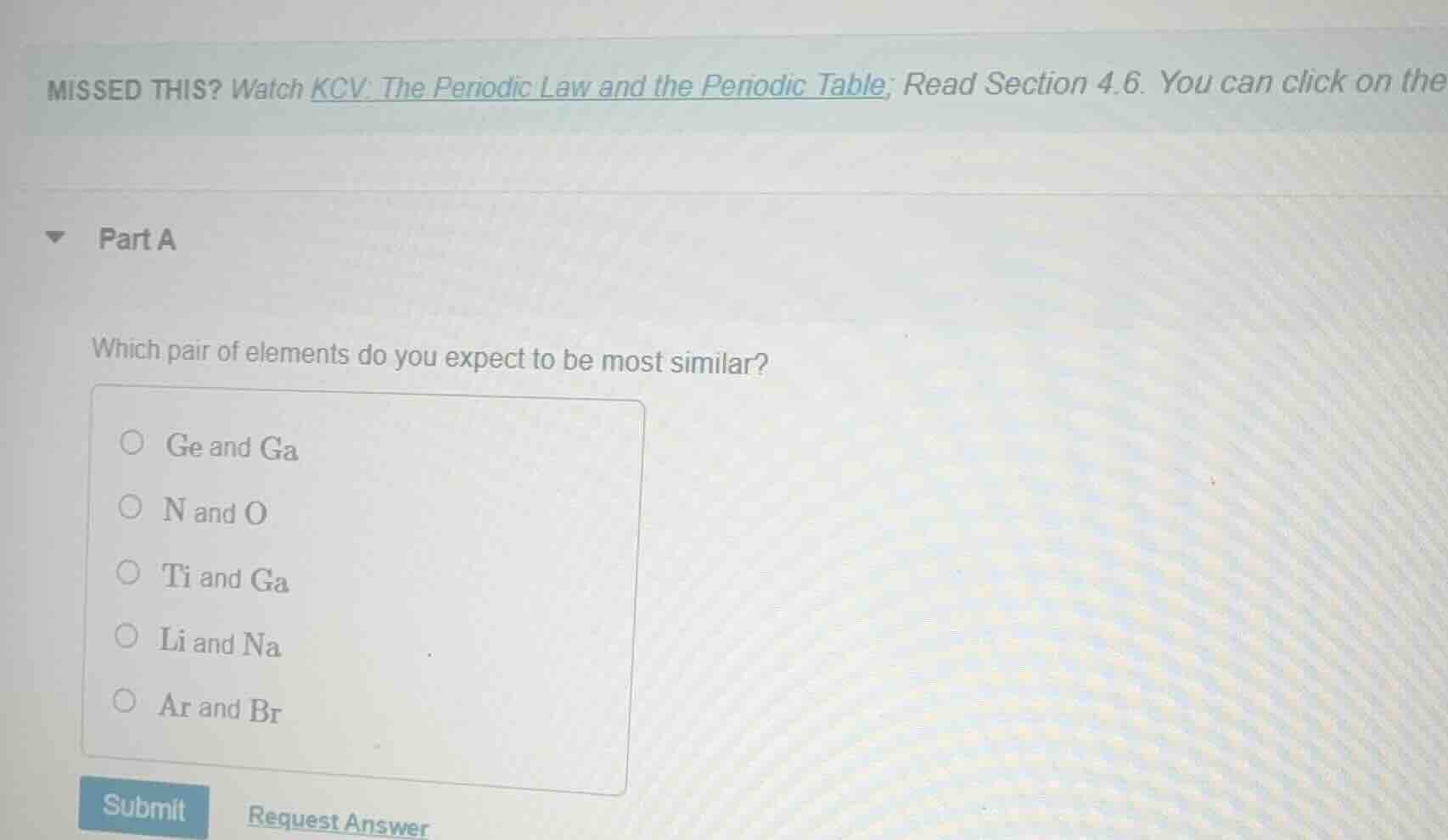
missed this? watch kcv: the periodic law and the periodic table; read section 4.6. you can click on the
part a
which pair of elements do you expect to be most similar?
○ ge and ga
○ n and o
○ ti and ga
○ li and na
○ ar and br
submit request answer
Brief Explanations
Elements in the same group (vertical column) of the periodic table have the same number of valence electrons, leading to very similar chemical and physical properties.
- Ge and Ga are adjacent in period 4, different groups.
- N and O are adjacent in period 2, different groups.
- Ti and Ga are in period 4, different groups.
- Li and Na are both in Group 1 (alkali metals), same valence electron count.
- Ar and Br are in period 3, different groups (Ar is noble gas, Br is halogen).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Li and Na