QUESTION IMAGE
Question
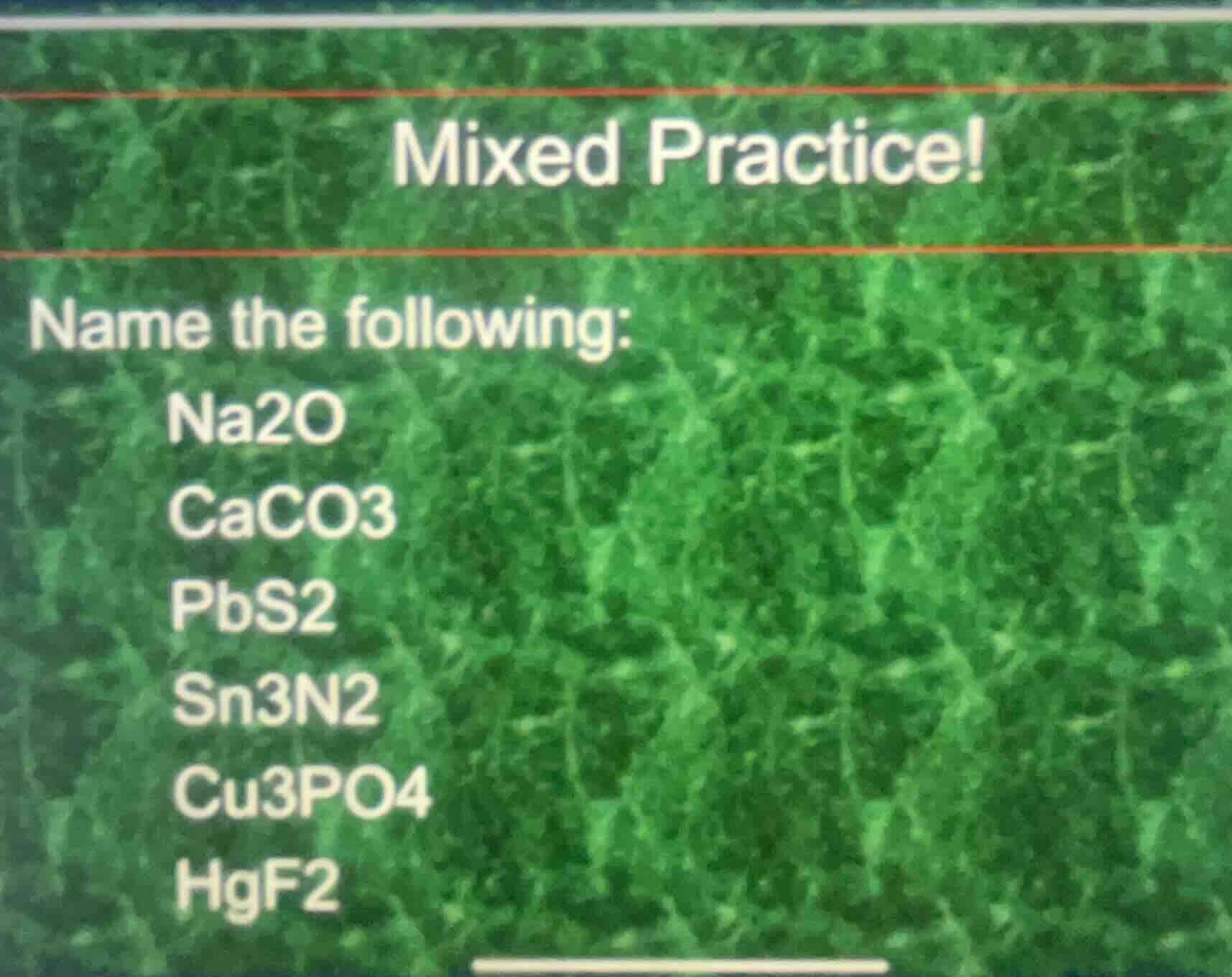
mixed practice!
name the following:
na2o
caco3
pbs2
sn3n2
cu3po4
hgf2
For $\boldsymbol{\ce{Na2O}}$:
Step1: Identify cation and anion
Cation: $\ce{Na+}$ (sodium ion), Anion: $\ce{O^{2-}}$ (oxide ion).
Step2: Name using ionic naming rules
Name the cation first, then the anion. The subscripts indicate the ratio (2 $\ce{Na+}$ to balance 1 $\ce{O^{2-}}$). So it's sodium oxide.
For $\boldsymbol{\ce{CaCO3}}$:
Step1: Identify cation and polyatomic anion
Cation: $\ce{Ca^{2+}}$ (calcium ion), Anion: $\ce{CO3^{2-}}$ (carbonate ion).
Step2: Name using ionic naming rules
Name cation then polyatomic anion. So it's calcium carbonate.
For $\boldsymbol{\ce{PbS2}}$:
Step1: Identify cation and anion
Cation: $\ce{Pb^{4+}}$ (lead(IV) ion, since 2 $\ce{S^{2-}}$ need 4+ from Pb: $x + 2\times(-2)=0\Rightarrow x = +4$), Anion: $\ce{S^{2-}}$ (sulfide ion).
Step2: Name using ionic naming rules (with Roman numeral for Pb's charge)
Lead(IV) sulfide.
For $\boldsymbol{\ce{Sn3N2}}$:
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
- $\ce{Na2O}$: Sodium Oxide
- $\ce{CaCO3}$: Calcium Carbonate
- $\ce{PbS2}$: Lead(IV) Sulfide
- $\ce{Sn3N2}$: Tin(II) Nitride
- $\ce{Cu3PO4}$: Copper(I) Phosphate
- $\ce{HgF2}$: Mercury(II) Fluoride