QUESTION IMAGE
Question
the modern atomic theory
quiz complete
1 2 3 4 5 6 7 8 9 10
time remain
58:02
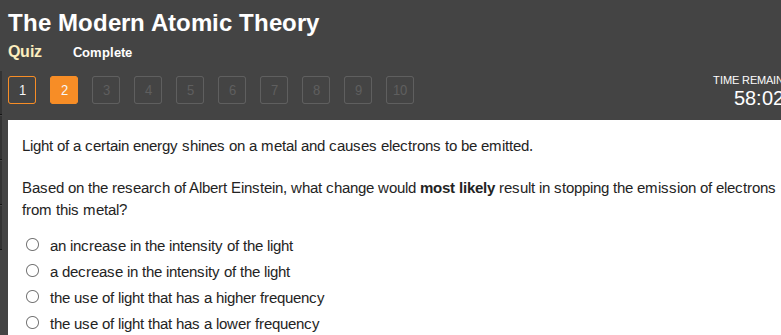
light of a certain energy shines on a metal and causes electrons to be emitted.
based on the research of albert einstein, what change would most likely result in stopping the emission of electrons from this metal?
○ an increase in the intensity of the light
○ a decrease in the intensity of the light
○ the use of light that has a higher frequency
○ the use of light that has a lower frequency
Einstein's photoelectric effect theory states that electrons are emitted from a metal only when the incident light has a frequency above the metal's threshold frequency. Lowering the frequency below this threshold will stop electron emission, regardless of light intensity. Higher frequency would increase electron energy, and intensity only affects the number of emitted electrons (if frequency is above threshold).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
the use of light that has a lower frequency