QUESTION IMAGE
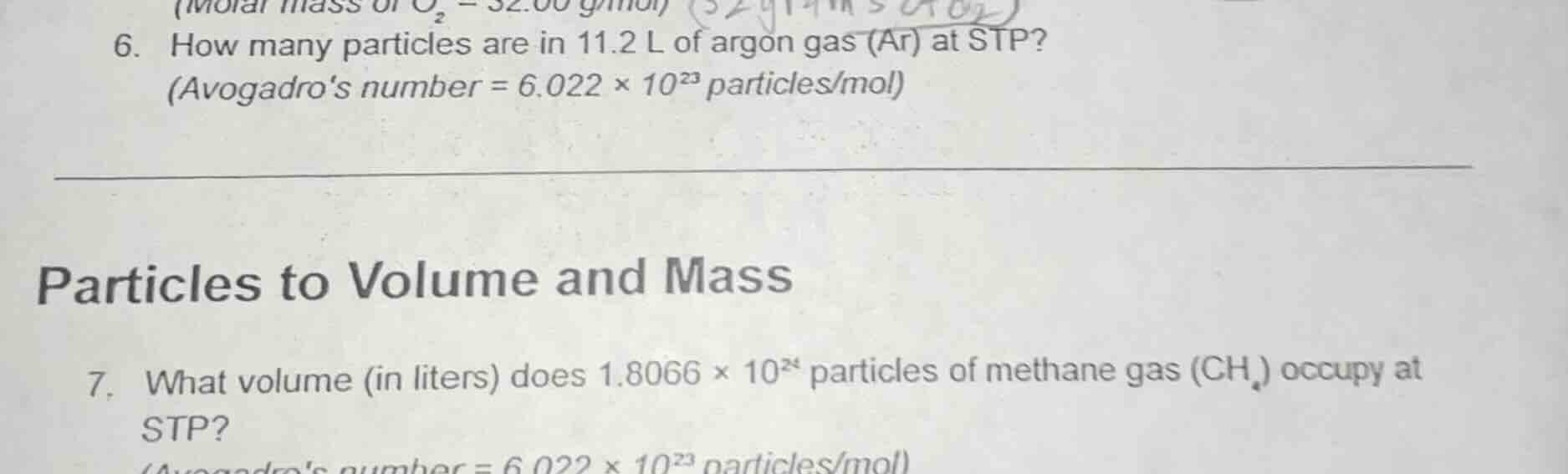
Question
(molar mass of o₂ = 32.00 g/mol)
- how many particles are in 11.2 l of argon gas (ar) at stp?
(avogadro’s number = 6.022 × 10²³ particles/mol)
particles to volume and mass
- what volume (in liters) does 1.8066 × 10²⁴ particles of methane gas (ch₄) occupy at stp?
(avogadro’s number = 6.022 × 10²³ particles/mol)
Question 6
Step1: Find moles of Ar at STP
At STP, 1 mole of any gas occupies 22.4 L. So moles of Ar, $n = \frac{V}{22.4\ L/mol}=\frac{11.2\ L}{22.4\ L/mol}=0.5\ mol$
Step2: Calculate number of particles
Using Avogadro's number, particles $= n\times N_A = 0.5\ mol\times6.022\times 10^{23}\ particles/mol = 3.011\times 10^{23}\ particles$
Step1: Find moles of $CH_4$
Moles, $n=\frac{\text{particles}}{N_A}=\frac{1.8066\times 10^{24}\ particles}{6.022\times 10^{23}\ particles/mol}=3\ mol$
Step2: Calculate volume at STP
At STP, volume $V = n\times22.4\ L/mol = 3\ mol\times22.4\ L/mol = 67.2\ L$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$3.011\times 10^{23}$ particles