QUESTION IMAGE
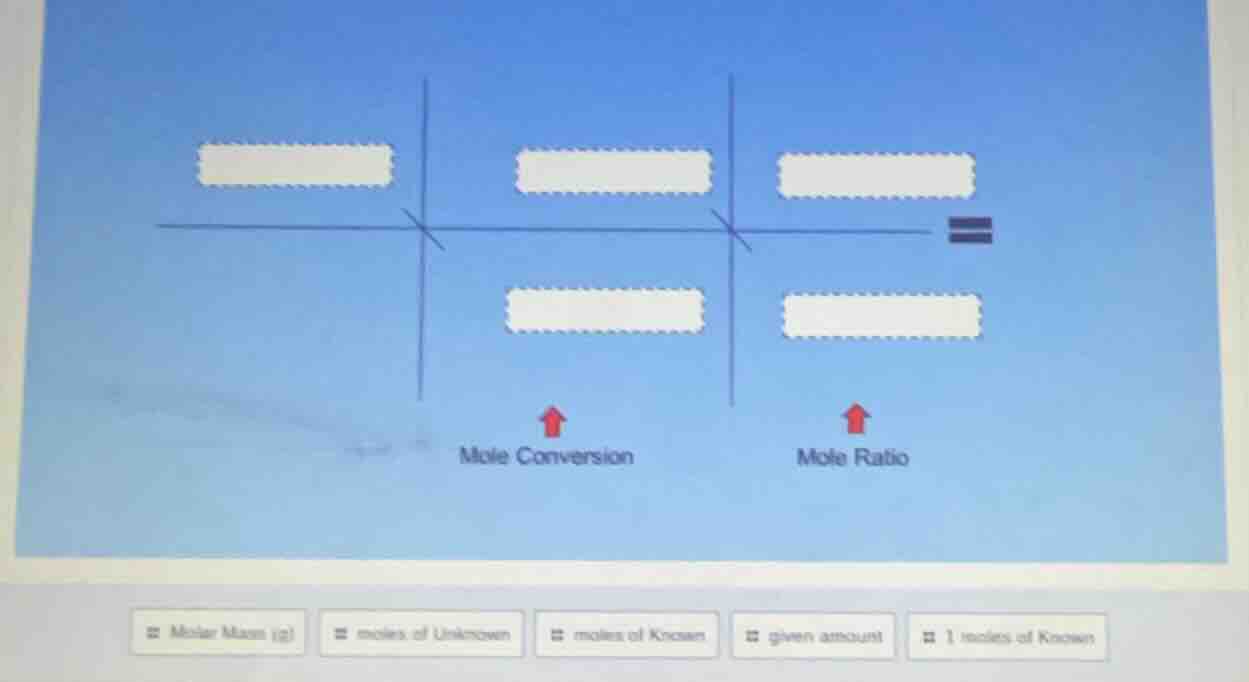
Question
mole conversion
mole ratio
molar mass (g)
moles of unknown
moles of known
given amount
1 moles of known
Step1: Map Mole Conversion components
The mole conversion step converts a given amount to moles of the known substance. This uses the given amount and the molar mass of the known (to convert mass to moles, for example).
Top left (input): $\text{given amount}$
Bottom middle (conversion factor): $\text{Molar Mass (g)}$
Top middle (output): $\text{moles of Known}$
Step2: Map Mole Ratio components
The mole ratio step converts moles of known to moles of unknown using the mole ratio from a balanced equation (1 mole of known relative to unknown).
Top middle (input): $\text{moles of Known}$
Bottom right (conversion factor): $\text{1 moles of Known}$
Top right (output): $\text{moles of Unknown}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Position | Filled Component |
|---|---|
| Bottom Middle | Molar Mass (g) |
| Top Middle | moles of Known |
| Bottom Right | 1 moles of Known |
| Top Right | moles of Unknown |
Full flow: $\text{given amount} \xrightarrow{\div \text{Molar Mass (g)}} \text{moles of Known} \xrightarrow{\times \frac{\text{moles of Unknown}}{\text{1 moles of Known}}} \text{moles of Unknown}$