QUESTION IMAGE
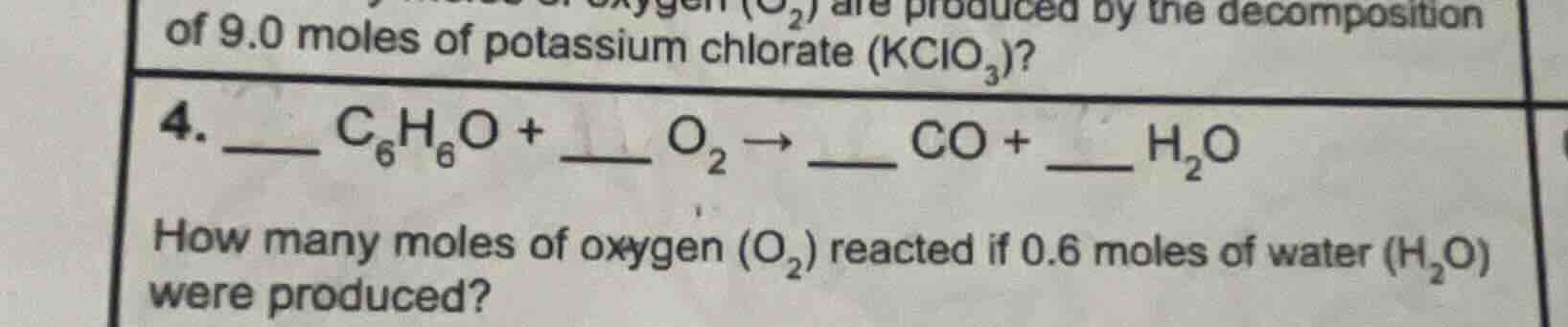
Question
of 9.0 moles of potassium chlorate (kclo₃)?
- __ c₆h₆o + o₂ → co + __ h₂o
how many moles of oxygen (o₂) reacted if 0.6 moles of water (h₂o) were produced?
Step1: Balance the chemical equation
First, balance the given reaction $\ce{C6H6O + O2 -> CO + H2O}$:
- Balance C: 6 C on left, so add 6 to $\ce{CO}$: $\ce{C6H6O + O2 -> 6CO + H2O}$
- Balance H: 6 H on left, so add 3 to $\ce{H2O}$: $\ce{C6H6O + O2 -> 6CO + 3H2O}$
- Balance O:
- Left O: $1 + 2x$ (from $\ce{C6H6O}$ and $\ce{O2}$)
- Right O: $6 + 3 = 9$
- Solve for $x$: $2x = 9 - 1 = 8 \implies x=4$
Balanced equation: $\ce{C6H6O + 4O2 -> 6CO + 3H2O}$
Step2: Set up mole ratio
From balanced equation, mole ratio of $\ce{O2}$ to $\ce{H2O}$ is $\frac{4\ \text{mol}\ \ce{O2}}{3\ \text{mol}\ \ce{H2O}}$
Step3: Calculate moles of $\ce{O2}$
Use given 0.6 mol $\ce{H2O}$ to find $\ce{O2}$:
$\text{Moles of }\ce{O2} = 0.6\ \text{mol}\ \ce{H2O} \times \frac{4\ \text{mol}\ \ce{O2}}{3\ \text{mol}\ \ce{H2O}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.8 moles of $\ce{O2}$ reacted.