QUESTION IMAGE
Question
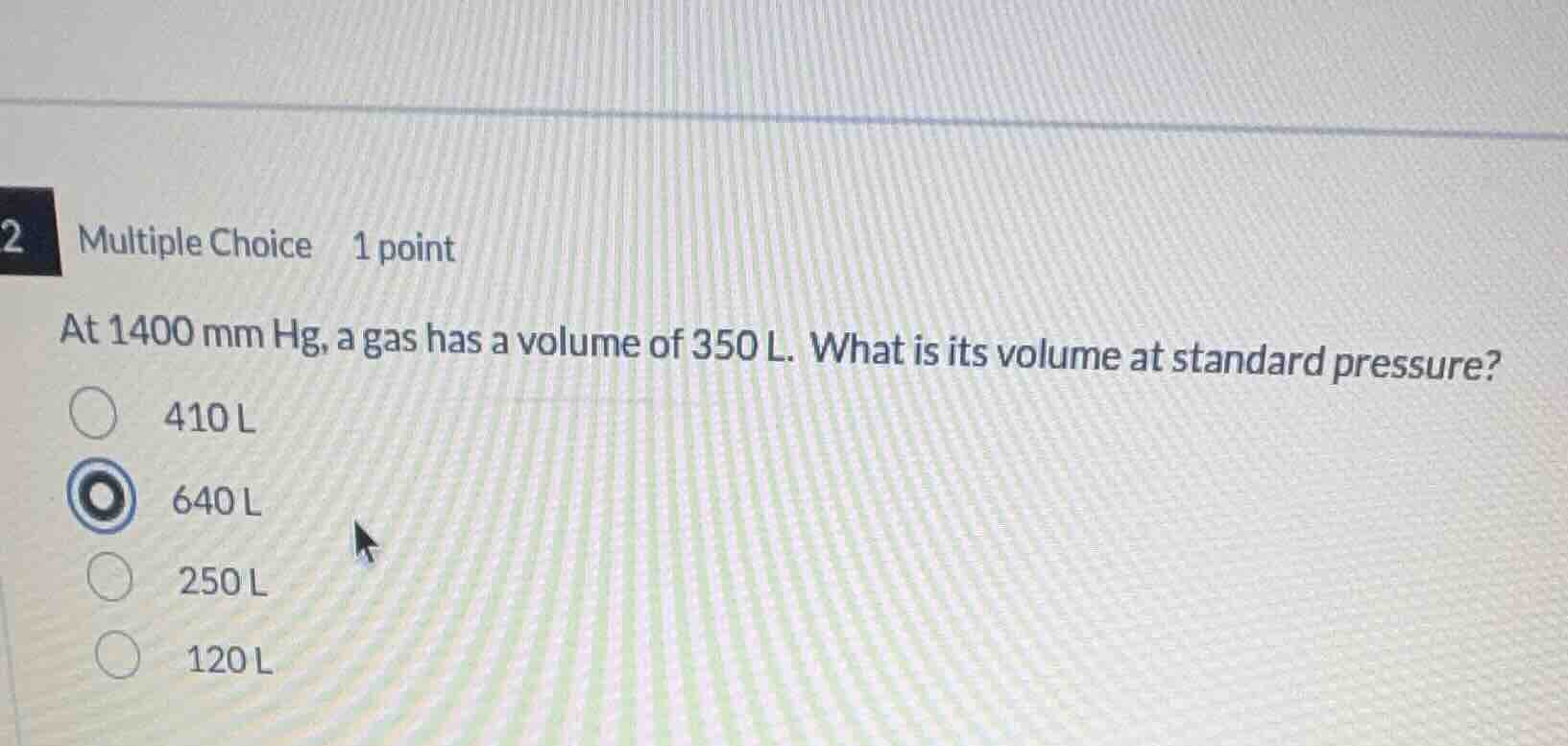
2 multiple choice 1 point
at 1400 mm hg, a gas has a volume of 350 l. what is its volume at standard pressure?
410 l
640 l
250 l
120 l
Step1: Recall Boyle's Law
$P_1V_1 = P_2V_2$
Step2: Define known values
$P_1 = 1400\ \text{mm Hg}$, $V_1 = 350\ \text{L}$, $P_2 = 760\ \text{mm Hg}$ (standard pressure)
Step3: Solve for $V_2$
$V_2 = \frac{P_1V_1}{P_2}$
Step4: Substitute values and calculate
$V_2 = \frac{1400 \times 350}{760} \approx 644.7\ \text{L}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
640 L