QUESTION IMAGE
Question
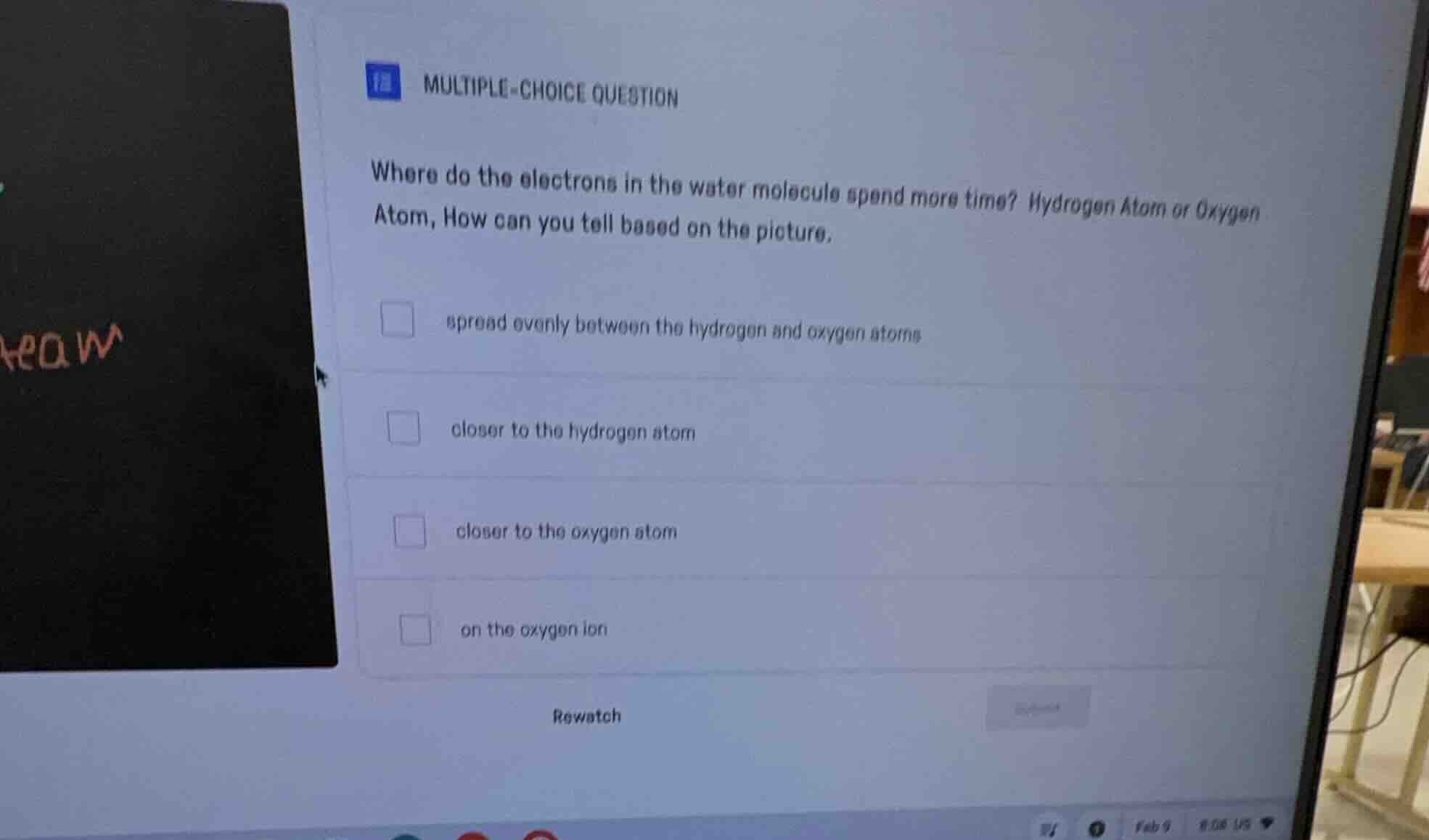
multiple-choice question
where do the electrons in the water molecule spend more time? hydrogen atom or oxygen atom, how can you tell based on the picture.
spread evenly between the hydrogen and oxygen atoms
closer to the hydrogen atom
closer to the oxygen atom
on the oxygen ion
rewatch
Oxygen has a higher electronegativity than hydrogen, meaning it attracts shared electrons more strongly. In a water molecule, this pulls the bonding electrons closer to the oxygen atom rather than hydrogen, and they do not spread evenly. There is no oxygen ion in a neutral water molecule.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
closer to the oxygen atom