QUESTION IMAGE
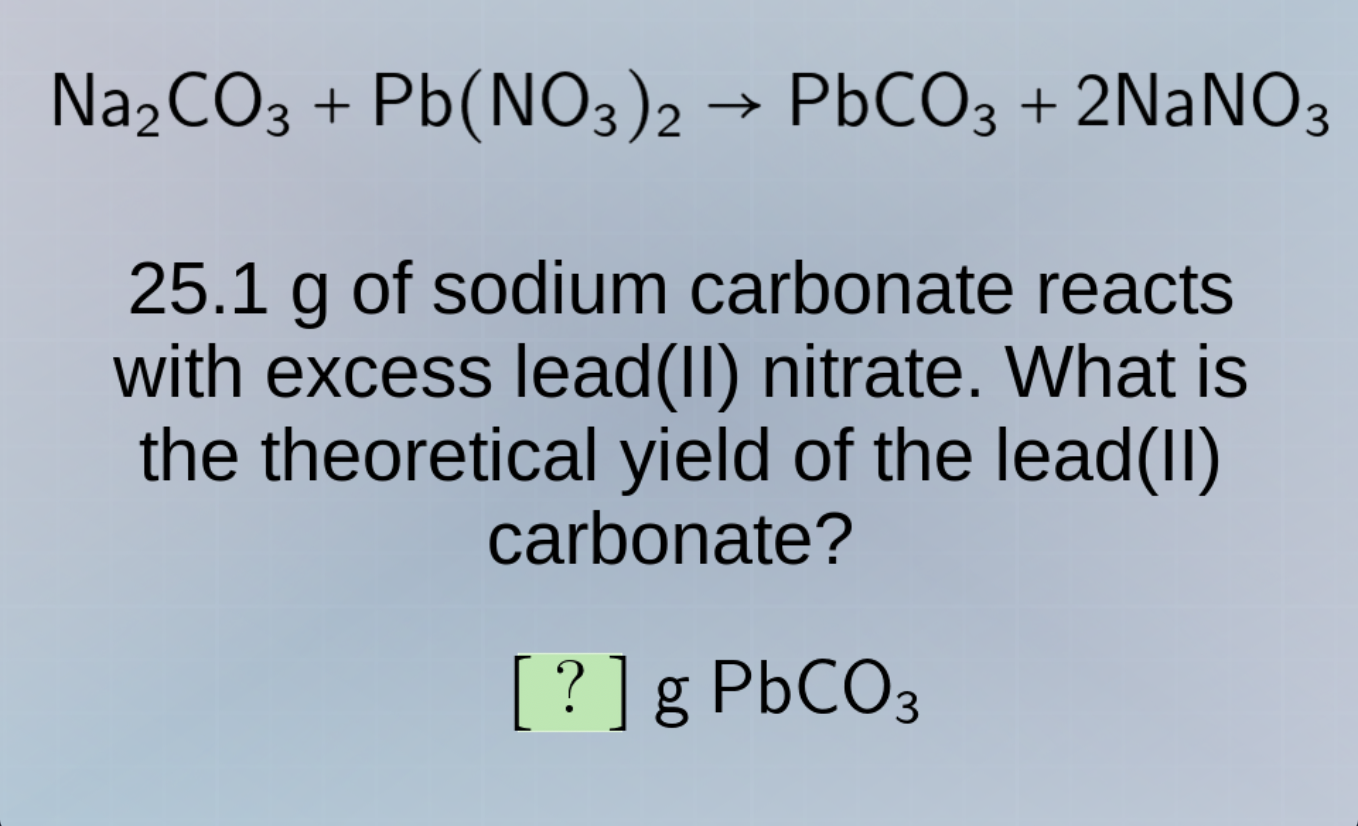
Question
na₂co₃ + pb(no₃)₂ → pbco₃ + 2nano₃
25.1 g of sodium carbonate reacts with excess lead(ii) nitrate. what is the theoretical yield of the lead(ii) carbonate?
? g pbco₃
Step1: Calculate moles of \( Na_2CO_3 \)
Molar mass of \( Na_2CO_3 \): \( 2\times23 + 12 + 3\times16 = 106 \, \text{g/mol} \)
Moles = \( \frac{\text{mass}}{\text{molar mass}} = \frac{25.1 \, \text{g}}{106 \, \text{g/mol}} \approx 0.2368 \, \text{mol} \)
Step2: Relate moles of \( Na_2CO_3 \) to \( PbCO_3 \)
From the balanced equation, \( 1 \, \text{mol} \, Na_2CO_3 \) produces \( 1 \, \text{mol} \, PbCO_3 \).
So, moles of \( PbCO_3 = 0.2368 \, \text{mol} \)
Step3: Calculate mass of \( PbCO_3 \)
Molar mass of \( PbCO_3 \): \( 207.2 + 12 + 3\times16 = 267.2 \, \text{g/mol} \)
Mass = \( \text{moles} \times \text{molar mass} = 0.2368 \, \text{mol} \times 267.2 \, \text{g/mol} \approx 63.3 \, \text{g} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 63.3 \)