QUESTION IMAGE
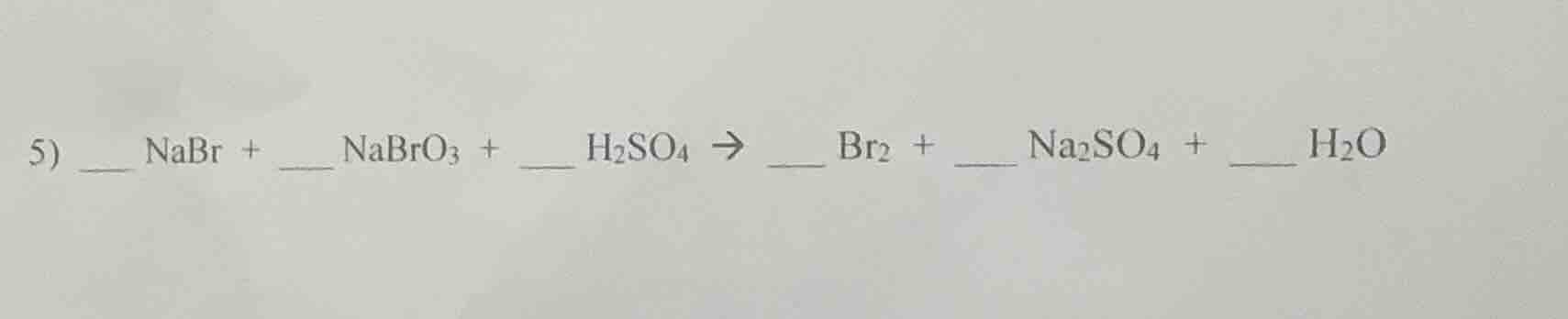
Question
- _ nabr + _ nabro₃ + _ h₂so₄ → _ br₂ + _ na₂so₄ + _ h₂o
Step1: Analyze Br oxidation states
In $\ce{NaBr}$, Br is -1. In $\ce{NaBrO3}$, Br is +5. In $\ce{Br2}$, Br is 0. The oxidation of Br⁻ (-1 to 0) and reduction of BrO₃⁻ (+5 to 0) occur. The number of electrons lost by Br⁻: 1 per atom. Electrons gained by BrO₃⁻: 5 per atom (from +5 to 0). To balance electrons, multiply $\ce{NaBr}$ by 5 and $\ce{NaBrO3}$ by 1. So tentative coefficients: 5 $\ce{NaBr}$, 1 $\ce{NaBrO3}$.
Step2: Balance Br atoms
Left Br: 5 (from $\ce{NaBr}$) + 1 (from $\ce{NaBrO3}$) = 6. So $\ce{Br2}$ coefficient: $\frac{6}{2}=3$. Now: 5 $\ce{NaBr}$ + 1 $\ce{NaBrO3}$ +... → 3 $\ce{Br2}$ +...
Step3: Balance Na atoms
Left Na: 5 + 1 = 6. So $\ce{Na2SO4}$ coefficient: $\frac{6}{2}=3$. Now: 5 $\ce{NaBr}$ + 1 $\ce{NaBrO3}$ +... → 3 $\ce{Br2}$ + 3 $\ce{Na2SO4}$ +...
Step4: Balance SO₄²⁻
$\ce{H2SO4}$ provides $\ce{SO4^{2-}}$. Since $\ce{Na2SO4}$ is 3, $\ce{H2SO4}$ coefficient is 3. Now: 5 $\ce{NaBr}$ + 1 $\ce{NaBrO3}$ + 3 $\ce{H2SO4}$ → 3 $\ce{Br2}$ + 3 $\ce{Na2SO4}$ +...
Step5: Balance H and O
$\ce{H2SO4}$ has 3×2 = 6 H, so $\ce{H2O}$ coefficient: 3 (since 3 $\ce{H2O}$ has 6 H). Check O: 3×4 (from $\ce{H2SO4}$) = 12. Right O: 3×4 (from $\ce{Na2SO4}$) + 3×1 (from $\ce{H2O}$) = 12 + 3? Wait, no: Wait, $\ce{Na2SO4}$ has 4 O per formula, 3×4=12. $\ce{H2O}$ has 1 O per formula, 3×1=3. Wait, left O: 3×4 (from $\ce{H2SO4}$) + 1×3 (from $\ce{NaBrO3}$) = 12 + 3 = 15? Wait, no, earlier step mistake. Wait, $\ce{NaBrO3}$ has 3 O. Let's recheck. Wait, after step 3, Na is 6, so $\ce{Na2SO4}$ is 3 (correct, 3×2=6 Na). $\ce{H2SO4}$: 3 (to get 3 $\ce{SO4^{2-}}$). Then O from $\ce{H2SO4}$: 3×4=12, O from $\ce{NaBrO3}$: 3. Total O left: 15. Right O: 3×4 (from $\ce{Na2SO4}$) + x×1 (from $\ce{H2O}$) = 12 + x. Also, H: 3×2=6, so $\ce{H2O}$ has 6 H, so $\ce{H2O}$ coefficient 3 (3×2=6 H). Then O right: 12 + 3 = 15, which matches left (12 + 3 = 15). Perfect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
5 $\ce{NaBr}$ + 1 $\ce{NaBrO3}$ + 3 $\ce{H2SO4}$ $
ightarrow$ 3 $\ce{Br2}$ + 3 $\ce{Na2SO4}$ + 3 $\ce{H2O}$
So coefficients: 5, 1, 3, 3, 3, 3.