QUESTION IMAGE
Question
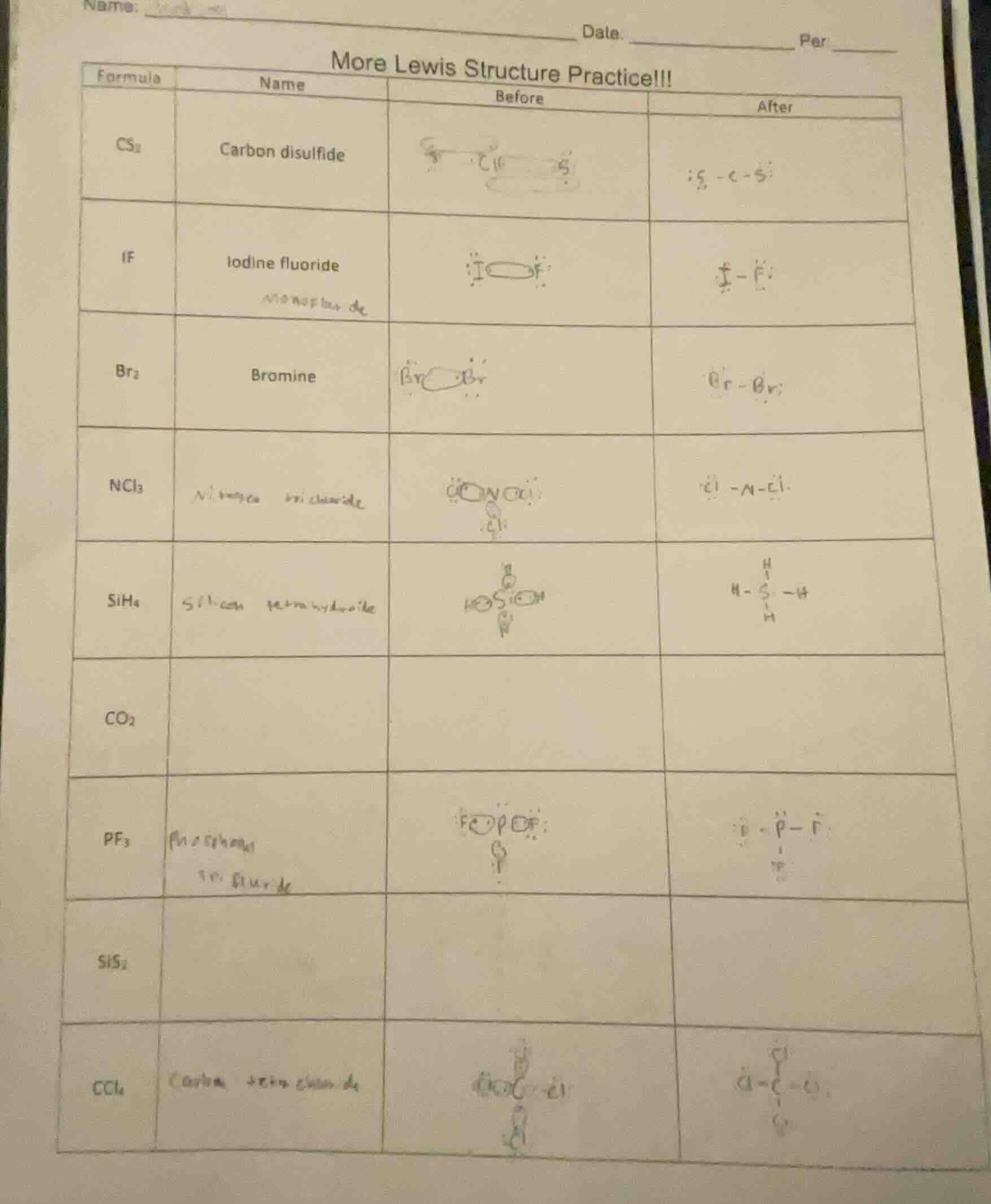
name. date. per more lewis structure practice!!! formula name before after cs₂ carbon disulfide if iodine fluoride br₂ bromine ncl₃ sih₄ co₂ pf₃ sis₂ ccl₄
To determine the Lewis structure of \( \ce{CO2} \) (Carbon Dioxide), we follow these steps:
Step 1: Calculate the total number of valence electrons
- Carbon (C) is in Group 14, so it has 4 valence electrons.
- Oxygen (O) is in Group 16, so each oxygen atom has 6 valence electrons. Since there are 2 oxygen atoms, the total for oxygen is \( 2 \times 6 = 12 \) valence electrons.
- The total number of valence electrons for \( \ce{CO2} \) is \( 4 + 12 = 16 \) valence electrons.
Step 2: Arrange the atoms
- Carbon is the central atom (less electronegative than oxygen), and the two oxygen atoms are bonded to it. The initial arrangement is \( \ce{O - C - O} \).
Step 3: Form single bonds and distribute remaining electrons
- Each single bond (C - O) uses 2 electrons. So, two single bonds use \( 2 \times 2 = 4 \) electrons.
- The remaining number of electrons is \( 16 - 4 = 12 \) electrons. These are distributed as lone pairs on the oxygen atoms. Each oxygen atom gets 6 electrons (3 lone pairs), but this would result in a structure where carbon only has 4 electrons (from the two single bonds), which is less than the octet. So, we need to form double bonds.
Step 4: Form double bonds to satisfy the octet rule
- To satisfy the octet for carbon, we convert one lone pair from each oxygen atom into a double bond. This gives us \( \ce{O = C = O} \).
- Now, let's check the number of electrons:
- Each double bond (C = O) has 4 electrons. Two double bonds have \( 2 \times 4 = 8 \) electrons.
- Each oxygen atom has 2 lone pairs (4 electrons) left. So, each oxygen has \( 4 + 4 = 8 \) electrons (octet), and carbon has \( 8 \) electrons (octet) from the two double bonds.
Step 5: Draw the Lewis structure
The Lewis structure of \( \ce{CO2} \) is:
\[
\ce{: \overset{..}{O} = C = \overset{..}{O} :}
\]
or in a more standard notation (with lone pairs shown):
\[
\ce{O::C::O}
\]
where each "::" represents a double bond (4 electrons) and the lone pairs on oxygen are also accounted for (each oxygen has 2 lone pairs, which can be written as \( \ce{:O::C::O:} \) with the lone pairs on the ends).
So, the Lewis structure of \( \ce{CO2} \) is \( \boldsymbol{\ce{O::C::O}} \) (or with lone pairs explicitly: \( \boldsymbol{\ce{: \overset{..}{O} = C = \overset{..}{O} :}} \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine the Lewis structure of \( \ce{CO2} \) (Carbon Dioxide), we follow these steps:
Step 1: Calculate the total number of valence electrons
- Carbon (C) is in Group 14, so it has 4 valence electrons.
- Oxygen (O) is in Group 16, so each oxygen atom has 6 valence electrons. Since there are 2 oxygen atoms, the total for oxygen is \( 2 \times 6 = 12 \) valence electrons.
- The total number of valence electrons for \( \ce{CO2} \) is \( 4 + 12 = 16 \) valence electrons.
Step 2: Arrange the atoms
- Carbon is the central atom (less electronegative than oxygen), and the two oxygen atoms are bonded to it. The initial arrangement is \( \ce{O - C - O} \).
Step 3: Form single bonds and distribute remaining electrons
- Each single bond (C - O) uses 2 electrons. So, two single bonds use \( 2 \times 2 = 4 \) electrons.
- The remaining number of electrons is \( 16 - 4 = 12 \) electrons. These are distributed as lone pairs on the oxygen atoms. Each oxygen atom gets 6 electrons (3 lone pairs), but this would result in a structure where carbon only has 4 electrons (from the two single bonds), which is less than the octet. So, we need to form double bonds.
Step 4: Form double bonds to satisfy the octet rule
- To satisfy the octet for carbon, we convert one lone pair from each oxygen atom into a double bond. This gives us \( \ce{O = C = O} \).
- Now, let's check the number of electrons:
- Each double bond (C = O) has 4 electrons. Two double bonds have \( 2 \times 4 = 8 \) electrons.
- Each oxygen atom has 2 lone pairs (4 electrons) left. So, each oxygen has \( 4 + 4 = 8 \) electrons (octet), and carbon has \( 8 \) electrons (octet) from the two double bonds.
Step 5: Draw the Lewis structure
The Lewis structure of \( \ce{CO2} \) is:
\[
\ce{: \overset{..}{O} = C = \overset{..}{O} :}
\]
or in a more standard notation (with lone pairs shown):
\[
\ce{O::C::O}
\]
where each "::" represents a double bond (4 electrons) and the lone pairs on oxygen are also accounted for (each oxygen has 2 lone pairs, which can be written as \( \ce{:O::C::O:} \) with the lone pairs on the ends).
So, the Lewis structure of \( \ce{CO2} \) is \( \boldsymbol{\ce{O::C::O}} \) (or with lone pairs explicitly: \( \boldsymbol{\ce{: \overset{..}{O} = C = \overset{..}{O} :}} \)).