QUESTION IMAGE
Question
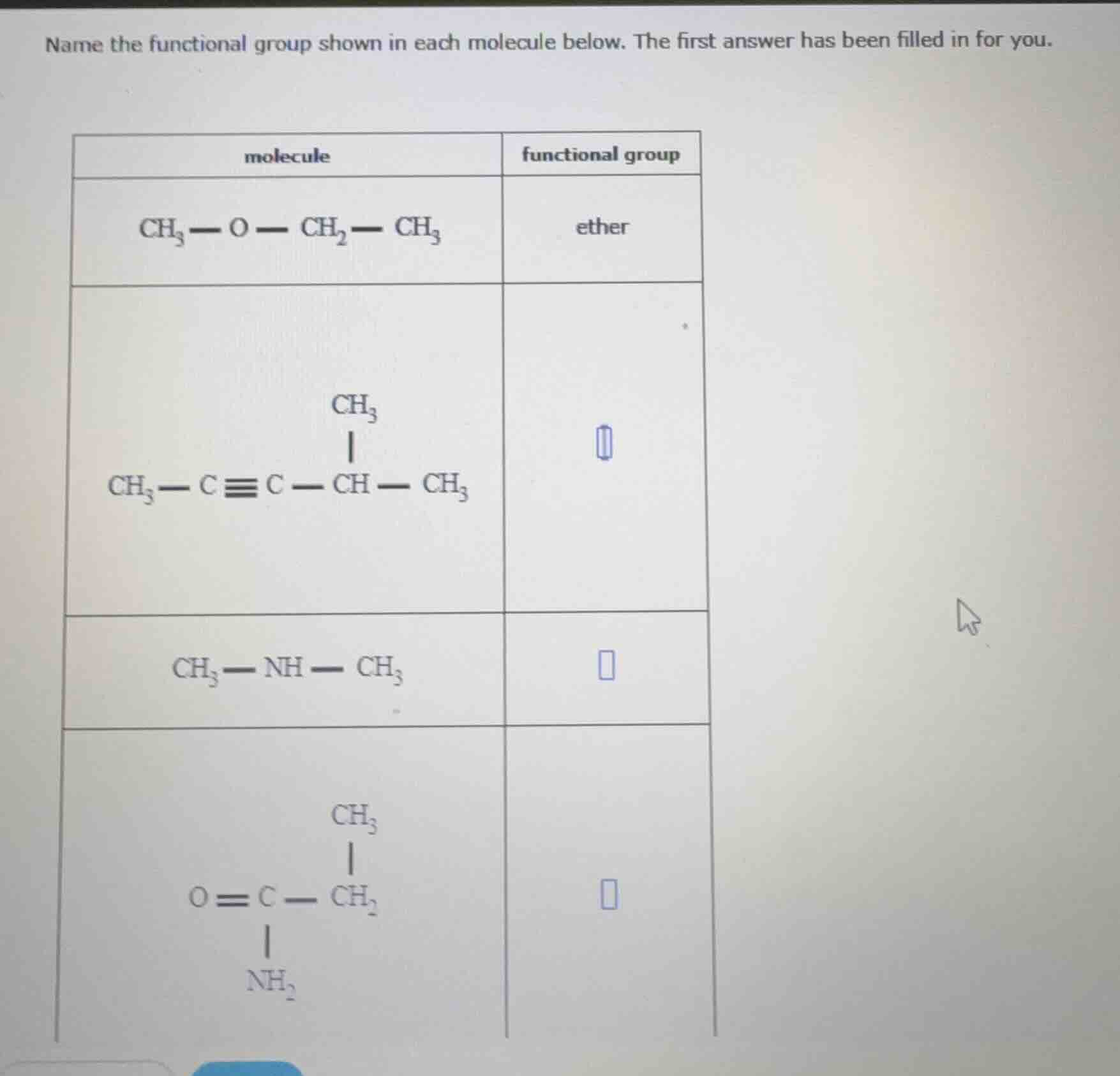
name the functional group shown in each molecule below. the first answer has been filled in for you.
| molecule | functional group |
|---|---|
| $\ce{ch_{3} - c \equiv c - ch(ch_{3}) - ch_{3}}$ | |
| $\ce{ch_{3} - nh - ch_{3}}$ | |
| $\ce{o = c(nh_{2}) - ch_{2}(ch_{3})}$ |
Second Molecule (Alkyne)
Step1: Identify the functional group
The molecule has a triple bond between two carbon atoms ($\ce{C\equiv C}$), which is characteristic of an alkyne functional group.
Step2: Name the functional group
Based on the presence of the carbon - carbon triple bond, the functional group is an alkyne (specifically an internal alkyne here).
Step1: Identify the functional group
The molecule has a $\ce{-NH -}$ group (with two alkyl groups attached to the nitrogen atom), which is the characteristic structure of a secondary amine. The general structure of an amine is $\ce{R - NH - R'}$ (for secondary amine), $\ce{R - NH2}$ (primary) or $\ce{R3N}$ (tertiary). Here, we have two methyl groups ($\ce{CH3 -}$) attached to the $\ce{-NH -}$ group, so it's an amine (secondary amine).
Step2: Name the functional group
From the structure $\ce{CH3 - NH - CH3}$, the functional group is an amine (more precisely, a secondary amine, but the general functional group category is amine).
Step1: Identify the functional group
The molecule has a $\ce{-CONH2}$ (or $\ce{-CO - NH2}$) group. The structure shows a carbonyl group ($\ce{C = O}$) bonded to an amino group ($\ce{-NH2}$), which is the characteristic structure of an amide. The general formula for an amide is $\ce{R - CONH2}$ (primary amide), $\ce{R - CONHR'}$ (secondary) or $\ce{R - CONR'2}$ (tertiary). Here, it's a primary amide with the structure $\ce{O = C(NH2)-CH2 - CH3}$ (after considering the $\ce{CH3}$ and $\ce{CH2}$ groups).
Step2: Name the functional group
Based on the $\ce{-CONH2}$ (carbonyl - amino) structure, the functional group is an amide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
alkyne