QUESTION IMAGE
Question
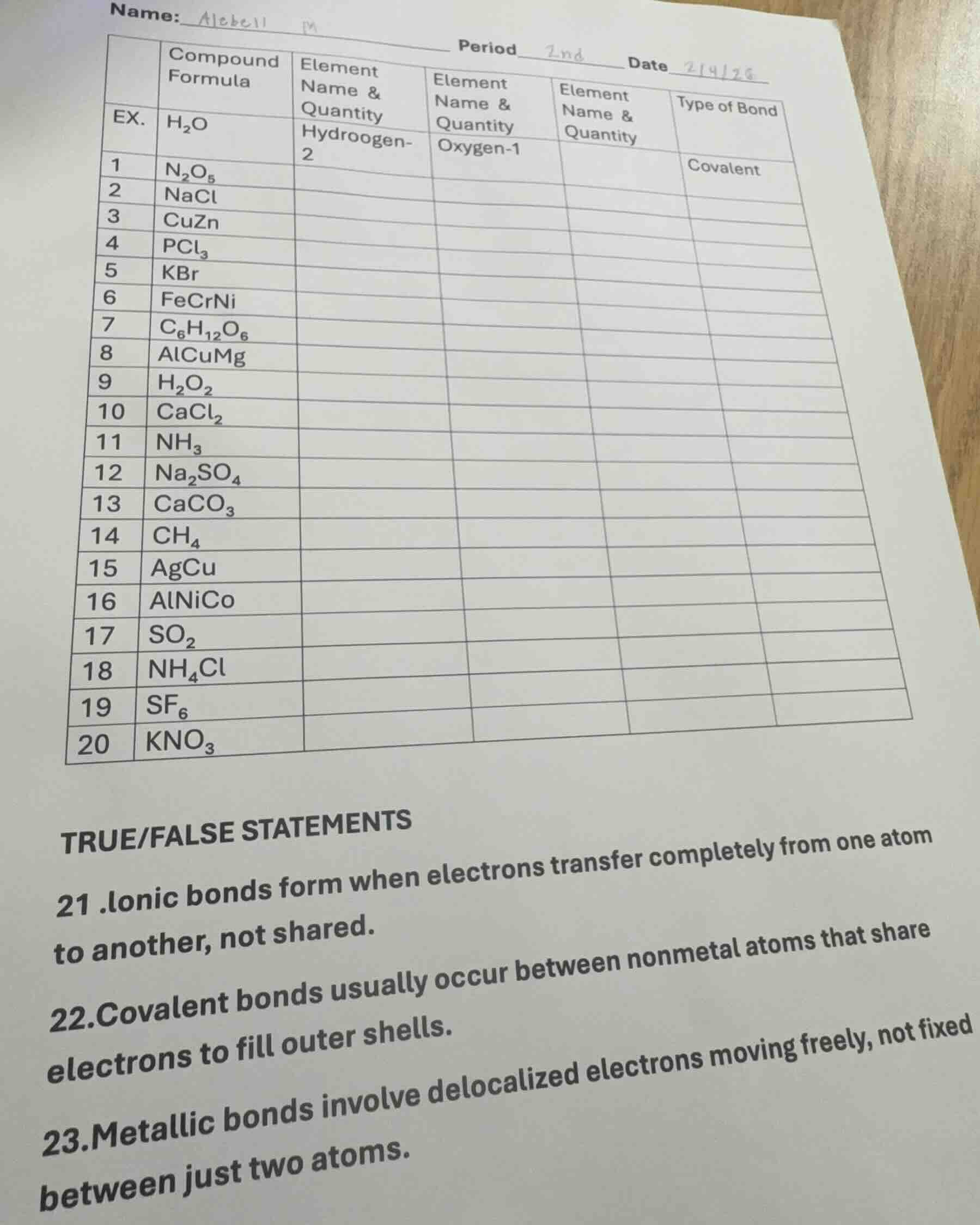
name:_____ period___ date_____
| compound formula | element name & quantity | element name & quantity | element name & quantity | type of bond | |
| ex. | h₂o | hydrooogen-2 | oxygen-1 | ||
| 1 | n₂o₅ | covalent | |||
| 2 | nacl | ||||
| 3 | cuzn | ||||
| 4 | pcl₃ | ||||
| 5 | kbr | ||||
| 6 | fecrni | ||||
| 7 | c₆h₁₂o₆ | ||||
| 8 | alcumg | ||||
| 9 | h₂o₂ | ||||
| 10 | cacl₂ | ||||
| 11 | nh₃ | ||||
| 12 | na₂so₄ | ||||
| 13 | caco₃ | ||||
| 14 | ch₄ | ||||
| 15 | agcu | ||||
| 16 | alnico | ||||
| 17 | so₂ | ||||
| 18 | nh₄cl | ||||
| 19 | sf₆ | ||||
| 20 | kno₃ |
true/false statements
- ionic bonds form when electrons transfer completely from one atom to another, not shared.
- covalent bonds usually occur between nonmetal atoms that share electrons to fill outer shells.
- metallic bonds involve delocalized electrons moving freely, not fixed between just two atoms.
Step1: Break down each compound
Extract elements and their counts from formulas, then classify bonds based on element types (metal-nonmetal = ionic; nonmetal-nonmetal = covalent; metal-metal = metallic; ionic + covalent = ionic with polyatomic ion).
Step2: Answer true/false questions
Apply definitions of ionic, covalent, metallic bonds.
---
Compound Table Completion:
| # | Compound Formula | Element Name & Quantity | Element Name & Quantity | Element Name & Quantity | Type of Bond |
|---|---|---|---|---|---|
| 2 | $\text{NaCl}$ | Sodium-1 | Chlorine-1 | - | Ionic |
| 3 | $\text{CuZn}$ | Copper-1 | Zinc-1 | - | Metallic |
| 4 | $\text{PCl}_3$ | Phosphorus-1 | Chlorine-3 | - | Covalent |
| 5 | $\text{KBr}$ | Potassium-1 | Bromine-1 | - | Ionic |
| 6 | $\text{FeCrNi}$ | Iron-1 | Chromium-1 | Nickel-1 | Metallic |
| 7 | $\text{C}_6\text{H}_{12}\text{O}_6$ | Carbon-6 | Hydrogen-12 | Oxygen-6 | Covalent |
| 8 | $\text{AlCuMg}$ | Aluminum-1 | Copper-1 | Magnesium-1 | Metallic |
| 9 | $\text{H}_2\text{O}_2$ | Hydrogen-2 | Oxygen-2 | - | Covalent |
| 10 | $\text{CaCl}_2$ | Calcium-1 | Chlorine-2 | - | Ionic |
| 11 | $\text{NH}_3$ | Nitrogen-1 | Hydrogen-3 | - | Covalent |
| 12 | $\text{Na}_2\text{SO}_4$ | Sodium-2 | Sulfur-1 | Oxygen-4 | Ionic (with covalent polyatomic ion) |
| 13 | $\text{CaCO}_3$ | Calcium-1 | Carbon-1 | Oxygen-3 | Ionic (with covalent polyatomic ion) |
| 14 | $\text{CH}_4$ | Carbon-1 | Hydrogen-4 | - | Covalent |
| 15 | $\text{AgCu}$ | Silver-1 | Copper-1 | - | Metallic |
| 16 | $\text{AlNiCo}$ | Aluminum-1 | Nickel-1 | Cobalt-1 | Metallic |
| 17 | $\text{SO}_2$ | Sulfur-1 | Oxygen-2 | - | Covalent |
| 18 | $\text{NH}_4\text{Cl}$ | Nitrogen-1 | Hydrogen-4 | Chlorine-1 | Ionic (with covalent polyatomic ion) |
| 19 | $\text{SF}_6$ | Sulfur-1 | Fluorine-6 | - | Covalent |
| 20 | $\text{KNO}_3$ | Potassium-1 | Nitrogen-1 | Oxygen-3 | Ionic (with covalent polyatomic ion) |
True/False Statements:
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- True
- True
- True