QUESTION IMAGE
Question
name:
q3 chemistry: u5l3 vsepr theory day 2
date:
practice set 2
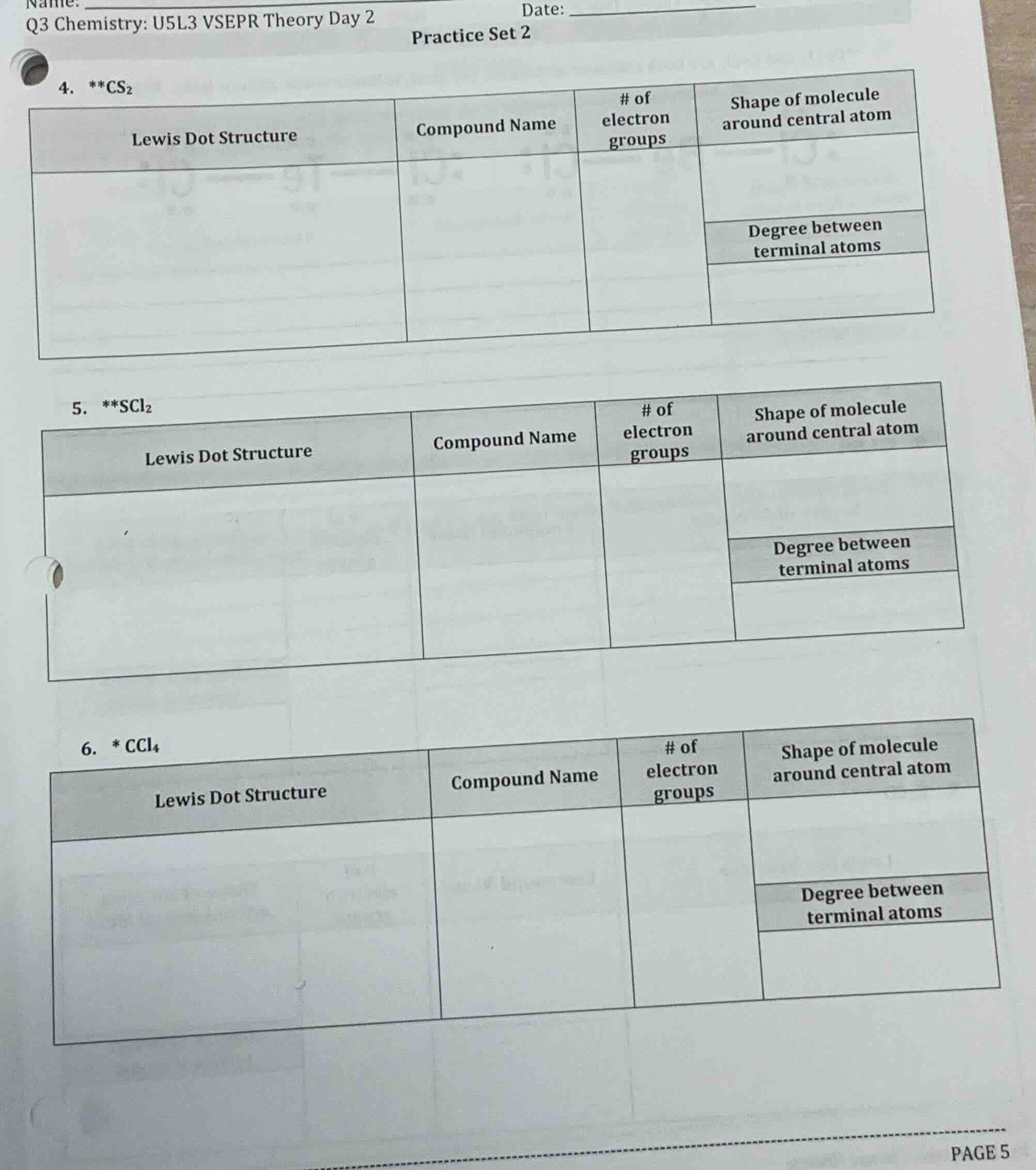
- cs₂
| lewis dot structure | compound name | # of electron groups | shape of molecule around central atom |
|---|---|---|---|
| degree between terminal atoms | |||
- scl₂
| lewis dot structure | compound name | # of electron groups | shape of molecule around central atom |
|---|---|---|---|
| degree between terminal atoms | |||
- * ccl₄
| lewis dot structure | compound name | # of electron groups | shape of molecule around central atom |
|---|---|---|---|
| degree between terminal atoms | |||
page 5
Problem 4: $\boldsymbol{CS_2}$
Step 1: Lewis Dot Structure
Carbon (C) has 4 valence electrons, sulfur (S) has 6. In $CS_2$, C is central. It forms double bonds with two S atoms:
$$\text{S}::\text{C}::\text{S}$$ (each S has 2 lone pairs, C has no lone pairs).
Step 2: Compound Name
Binary molecular compound: name first element (carbon), second (sulfur) with -ide suffix. So carbon disulfide.
Step 3: # of Electron Groups
Electron groups = bonding groups (no lone pairs on C). 2 double bonds → 2 electron groups.
Step 4: Molecular Shape
2 electron groups, no lone pairs → linear (VSEPR: $AX_2$).
Step 5: Bond Angle
Linear shape → bond angle = $180^\circ$.
Problem 5: $\boldsymbol{SCl_2}$
Step 1: Lewis Dot Structure
Sulfur (S) has 6 valence electrons, Cl has 7. S is central, bonded to 2 Cl atoms, with 2 lone pairs on S:
$$\text{Cl}-\text{S}-\text{Cl}$$ (each Cl has 3 lone pairs, S has 2 lone pairs).
Step 2: Compound Name
Binary molecular compound: sulfur dichloride.
Step 3: # of Electron Groups
Bonding groups (2) + lone pairs (2) → 4 electron groups.
Step 4: Molecular Shape
4 electron groups (2 bonding, 2 lone pairs) → bent (VSEPR: $AX_2E_2$).
Step 5: Bond Angle
Lone pairs repel, so angle < $109.5^\circ$ (approx $103^\circ$ for $SCl_2$).
Problem 6: $\boldsymbol{CCl_4}$
Step 1: Lewis Dot Structure
Carbon (C) has 4 valence electrons, Cl has 7. C is central, bonded to 4 Cl atoms (single bonds), no lone pairs on C:
$$\text{Cl}-\text{C}-\text{Cl}$$ (each Cl has 3 lone pairs, C has no lone pairs, 4 single bonds).
Step 2: Compound Name
Binary molecular compound: carbon tetrachloride.
Step 3: # of Electron Groups
4 single bonds → 4 electron groups (no lone pairs on C).
Step 4: Molecular Shape
4 electron groups, no lone pairs → tetrahedral (VSEPR: $AX_4$).
Step 5: Bond Angle
Tetrahedral shape → bond angle = $109.5^\circ$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
4. $CS_2$
- Lewis Dot Structure: $\boldsymbol{\text{S}::\text{C}::\text{S}}$ (S: 2 lone pairs each, C: 0)
- Compound Name: $\boldsymbol{\text{carbon disulfide}}$
- # of electron groups: $\boldsymbol{2}$
- Shape: $\boldsymbol{\text{linear}}$
- Bond angle: $\boldsymbol{180^\circ}$
5. $SCl_2$
- Lewis Dot Structure: $\boldsymbol{\text{Cl}-\text{S}-\text{Cl}}$ (Cl: 3 lone pairs each, S: 2 lone pairs)
- Compound Name: $\boldsymbol{\text{sulfur dichloride}}$
- # of electron groups: $\boldsymbol{4}$
- Shape: $\boldsymbol{\text{bent}}$
- Bond angle: $\boldsymbol{\approx 103^\circ}$ (or $< 109.5^\circ$)
6. $CCl_4$
- Lewis Dot Structure: $\boldsymbol{\text{Cl}-\text{C}-\text{Cl}}$ (4 single bonds, Cl: 3 lone pairs each, C: 0)
- Compound Name: $\boldsymbol{\text{carbon tetrachloride}}$
- # of electron groups: $\boldsymbol{4}$
- Shape: $\boldsymbol{\text{tetrahedral}}$
- Bond angle: $\boldsymbol{109.5^\circ}$