QUESTION IMAGE
Question
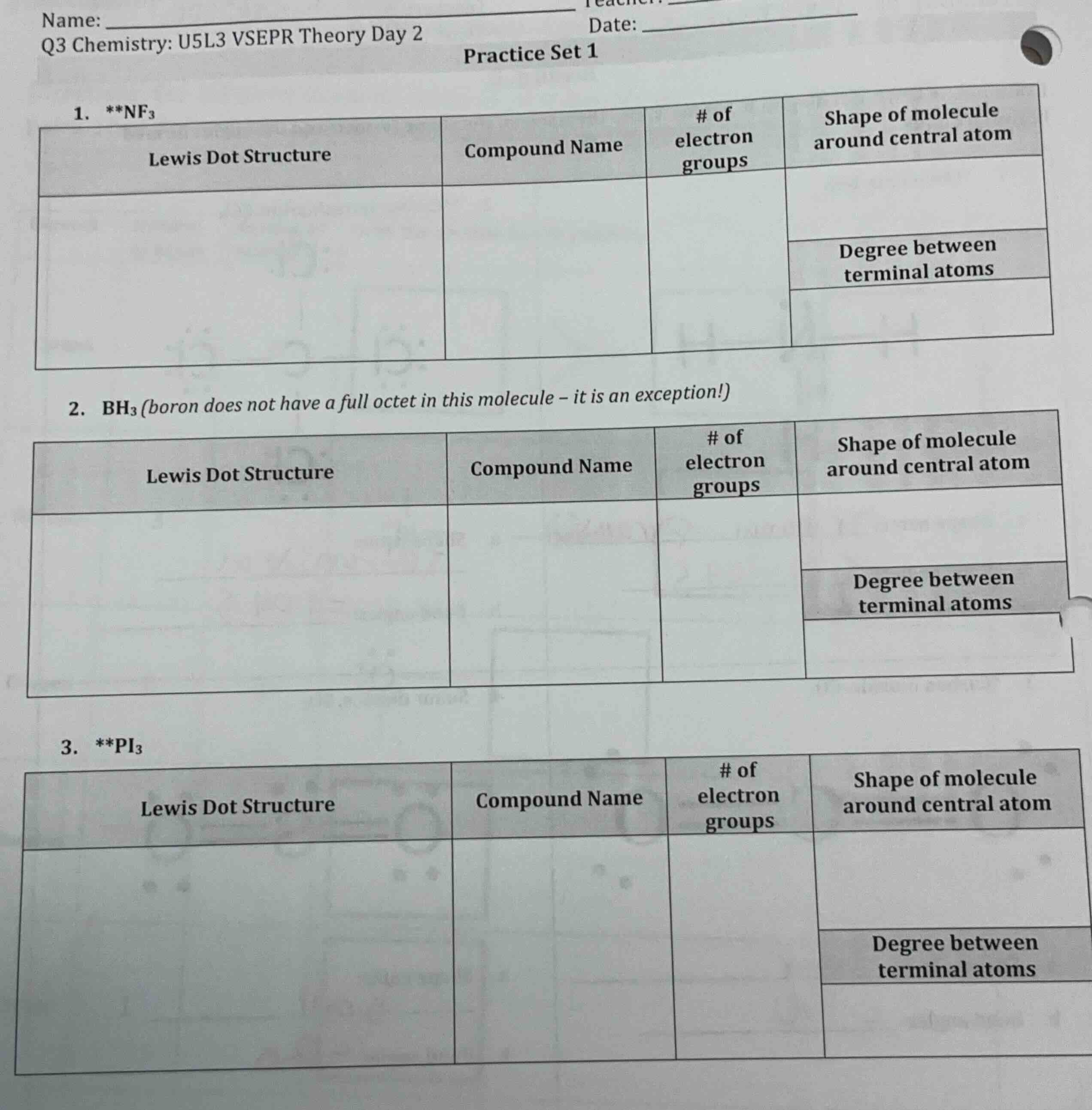
name: ______
q3 chemistry: u5l3 vsepr theory day 2
date: ______
practice set 1
- nf₃
| lewis dot structure | compound name | # of electron groups | shape of molecule around central atom |
|---|---|---|---|
| degree between terminal atoms | |||
- bh₃ (boron does not have a full octet in this molecule – it is an exception!)
| lewis dot structure | compound name | # of electron groups | shape of molecule around central atom |
|---|---|---|---|
| degree between terminal atoms | |||
- pi₃
| lewis dot structure | compound name | # of electron groups | shape of molecule around central atom |
|---|---|---|---|
| degree between terminal atoms | |||
Problem 1: $\boldsymbol{NF_3}$
Step 1: Lewis Dot Structure
Nitrogen (N) is central, with 5 valence electrons. Each Fluorine (F) has 7 valence electrons. N forms 3 single bonds with F (sharing 1 e⁻ each), and has 1 lone pair (2 e⁻). So:
$\ce{N}$ (central) with 3 $\ce{F}$ atoms (each with 3 lone pairs) and 1 lone pair on $\ce{N}$.
Step 2: Compound Name
Named as Nitrogen Trifluoride.
Step 3: # of Electron Groups
Electron groups = bonding groups + lone pairs. 3 bonding (N - F) + 1 lone pair = 4.
Step 4: Molecular Shape (VSEPR)
4 electron groups (3 bonding, 1 lone pair) → Trigonal Pyramidal.
Step 5: Bond Angle
Lone pair repulsion compresses the angle from 109.5° (tetrahedral) to ~107°.
Problem 2: $\boldsymbol{BH_3}$
Step 1: Lewis Dot Structure
Boron (B) is central (3 valence e⁻). 3 single bonds with H (each H has 1 e⁻). B has no lone pairs (exception to octet rule).
Step 2: Compound Name
Boron Trihydride (or Borane).
Step 3: # of Electron Groups
3 bonding groups (B - H) + 0 lone pairs = 3.
Step 4: Molecular Shape (VSEPR)
3 electron groups (all bonding) → Trigonal Planar.
Step 5: Bond Angle
Trigonal planar angle = 120°.
Problem 3: $\boldsymbol{PI_3}$
Step 1: Lewis Dot Structure
Phosphorus (P) is central (5 valence e⁻). 3 single bonds with I (each I has 3 lone pairs), and 1 lone pair on P.
Step 2: Compound Name
Phosphorus Triiodide.
Step 3: # of Electron Groups
3 bonding (P - I) + 1 lone pair = 4.
Step 4: Molecular Shape (VSEPR)
4 electron groups (3 bonding, 1 lone pair) → Trigonal Pyramidal.
Step 5: Bond Angle
Lone pair repulsion → ~107° (similar to $\ce{NF_3}$).
Summary Table
| Compound | Lewis Dot Structure (Brief) | Compound Name | # Electron Groups | Molecular Shape | Bond Angle |
|---|---|---|---|---|---|
| $\ce{BH_3}$ | B - 3 H (no lone pairs on B) | Boron Trihydride | 3 | Trigonal Planar | 120° |
| $\ce{PI_3}$ | P (1 lone pair) - 3 I (3 lone pairs each) | Phosphorus Triiodide | 4 | Trigonal Pyramidal | ~107° |
Final Answers (Filled Table for Each Compound)
1. $\boldsymbol{NF_3}$
- Lewis Dot Structure: $\ce{N}$ (lone pair) bonded to 3 $\ce{F}$ (each with 3 lone pairs).
- Compound Name: Nitrogen Trifluoride
- # of Electron Groups: 4
- Shape: Trigonal Pyramidal
- Bond Angle: ~107°
2. $\boldsymbol{BH_3}$
- Lewis Dot Structure: $\ce{B}$ bonded to 3 $\ce{H}$ (no lone pairs on $\ce{B}$).
- Compound Name: Boron Trihydride
- # of Electron Groups: 3
- Shape: Trigonal Planar
- Bond Angle: 120°
3. $\boldsymbol{PI_3}$
- Lewis Dot Structure: $\ce{P}$ (lone pair) bonded to 3 $\ce{I}$ (each with 3 lone pairs).
- Compound Name: Phosphorus Triiodide
- # of Electron Groups: 4
- Shape: Trigonal Pyramidal
- Bond Angle: ~107°
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Problem 1: $\boldsymbol{NF_3}$
Step 1: Lewis Dot Structure
Nitrogen (N) is central, with 5 valence electrons. Each Fluorine (F) has 7 valence electrons. N forms 3 single bonds with F (sharing 1 e⁻ each), and has 1 lone pair (2 e⁻). So:
$\ce{N}$ (central) with 3 $\ce{F}$ atoms (each with 3 lone pairs) and 1 lone pair on $\ce{N}$.
Step 2: Compound Name
Named as Nitrogen Trifluoride.
Step 3: # of Electron Groups
Electron groups = bonding groups + lone pairs. 3 bonding (N - F) + 1 lone pair = 4.
Step 4: Molecular Shape (VSEPR)
4 electron groups (3 bonding, 1 lone pair) → Trigonal Pyramidal.
Step 5: Bond Angle
Lone pair repulsion compresses the angle from 109.5° (tetrahedral) to ~107°.
Problem 2: $\boldsymbol{BH_3}$
Step 1: Lewis Dot Structure
Boron (B) is central (3 valence e⁻). 3 single bonds with H (each H has 1 e⁻). B has no lone pairs (exception to octet rule).
Step 2: Compound Name
Boron Trihydride (or Borane).
Step 3: # of Electron Groups
3 bonding groups (B - H) + 0 lone pairs = 3.
Step 4: Molecular Shape (VSEPR)
3 electron groups (all bonding) → Trigonal Planar.
Step 5: Bond Angle
Trigonal planar angle = 120°.
Problem 3: $\boldsymbol{PI_3}$
Step 1: Lewis Dot Structure
Phosphorus (P) is central (5 valence e⁻). 3 single bonds with I (each I has 3 lone pairs), and 1 lone pair on P.
Step 2: Compound Name
Phosphorus Triiodide.
Step 3: # of Electron Groups
3 bonding (P - I) + 1 lone pair = 4.
Step 4: Molecular Shape (VSEPR)
4 electron groups (3 bonding, 1 lone pair) → Trigonal Pyramidal.
Step 5: Bond Angle
Lone pair repulsion → ~107° (similar to $\ce{NF_3}$).
Summary Table
| Compound | Lewis Dot Structure (Brief) | Compound Name | # Electron Groups | Molecular Shape | Bond Angle |
|---|---|---|---|---|---|
| $\ce{BH_3}$ | B - 3 H (no lone pairs on B) | Boron Trihydride | 3 | Trigonal Planar | 120° |
| $\ce{PI_3}$ | P (1 lone pair) - 3 I (3 lone pairs each) | Phosphorus Triiodide | 4 | Trigonal Pyramidal | ~107° |
Final Answers (Filled Table for Each Compound)
1. $\boldsymbol{NF_3}$
- Lewis Dot Structure: $\ce{N}$ (lone pair) bonded to 3 $\ce{F}$ (each with 3 lone pairs).
- Compound Name: Nitrogen Trifluoride
- # of Electron Groups: 4
- Shape: Trigonal Pyramidal
- Bond Angle: ~107°
2. $\boldsymbol{BH_3}$
- Lewis Dot Structure: $\ce{B}$ bonded to 3 $\ce{H}$ (no lone pairs on $\ce{B}$).
- Compound Name: Boron Trihydride
- # of Electron Groups: 3
- Shape: Trigonal Planar
- Bond Angle: 120°
3. $\boldsymbol{PI_3}$
- Lewis Dot Structure: $\ce{P}$ (lone pair) bonded to 3 $\ce{I}$ (each with 3 lone pairs).
- Compound Name: Phosphorus Triiodide
- # of Electron Groups: 4
- Shape: Trigonal Pyramidal
- Bond Angle: ~107°