QUESTION IMAGE
Question
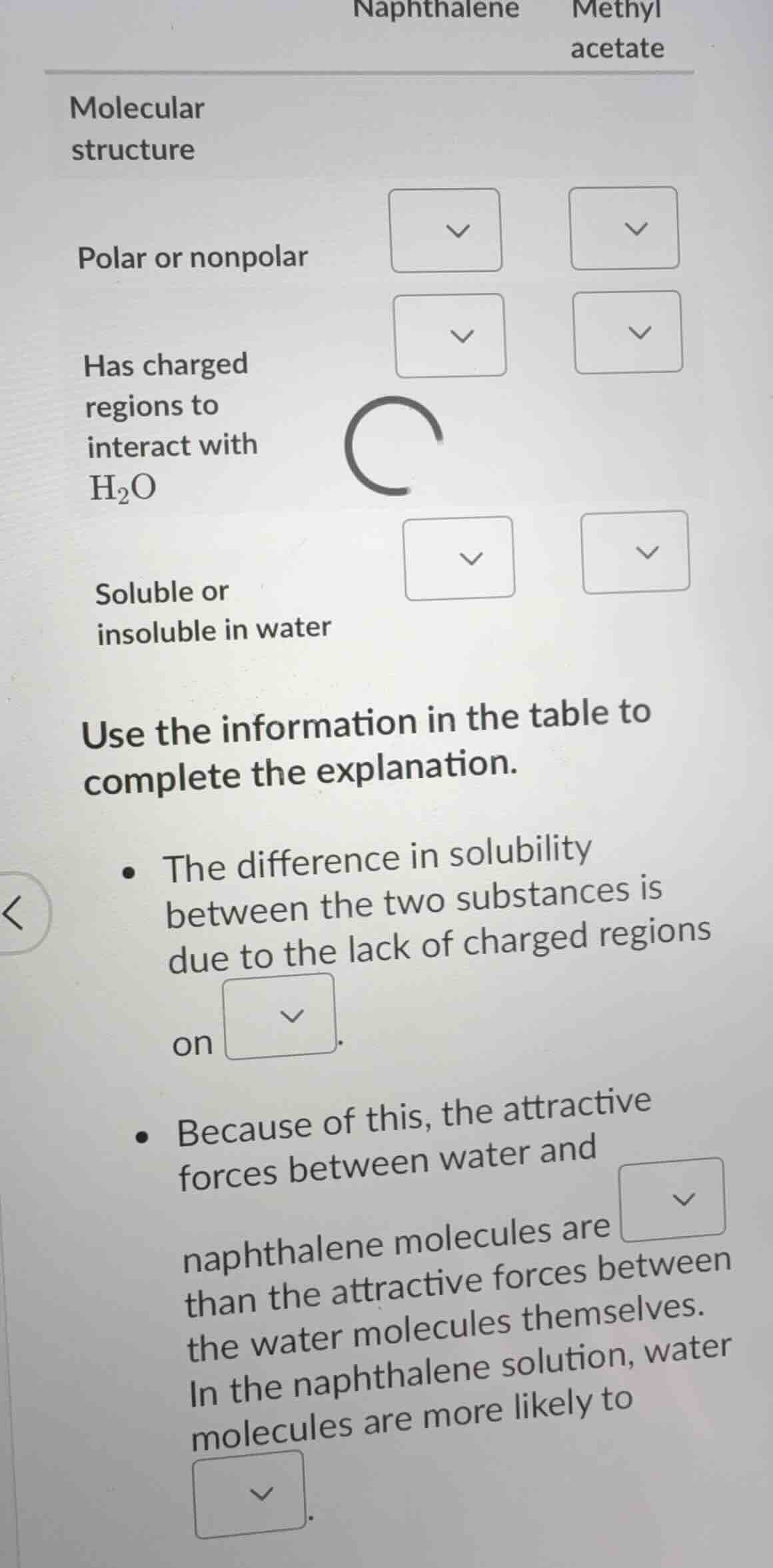
naphthalene methyl acetate
molecular structure
polar or nonpolar
has charged regions to interact with h₂o
soluble or insoluble in water
use the information in the table to complete the explanation.
- the difference in solubility between the two substances is due to the lack of charged regions on.
- because of this, the attractive forces between water and naphthalene molecules are than the attractive forces between the water molecules themselves. in the naphthalene solution, water molecules are more likely to.
To solve this, we analyze the properties of naphthalene and methyl acetate:
Step 1: Recall Solubility Principles
Water is polar ($\ce{H2O}$ has a bent structure, polar bonds, and a net dipole). Polar substances or those with charged regions (like $\ce{-OH}$, $\ce{-COOH}$, or ionic groups) dissolve in water (like dissolves like). Nonpolar substances (no net dipole, no charged regions) do not dissolve well in water.
Step 2: Analyze Naphthalene
Naphthalene ($\ce{C10H8}$) is a nonpolar molecule (consists of only carbon and hydrogen, with a symmetrical aromatic structure). It has no charged regions (no polar bonds with large electronegativity differences, no ions). Thus, it cannot interact favorably with polar water molecules.
Step 3: Analyze Methyl Acetate
Methyl acetate ($\ce{CH3COOCH3}$) has a polar carbonyl group ($\ce{C=O}$) and polar $\ce{C-O}$ bonds. It has charged regions (partial positive on $\ce{C}$ of $\ce{C=O}$, partial negative on $\ce{O}$ of $\ce{C=O}$) that can interact with water’s polar regions.
Step 4: Solubility Difference
The difference in solubility arises because naphthalene lacks charged regions. Attractive forces between water and naphthalene are weaker than water-water forces (since naphthalene is nonpolar). In a naphthalene “solution” (actually insoluble), water molecules prefer to stay with other water molecules (so naphthalene is insoluble, and water molecules “stay together” or “exclude” naphthalene).
Filling the Blanks:
- “The difference in solubility between the two substances is due to the lack of charged regions on $\boldsymbol{\text{naphthalene}}$.”
- “Because of this, the attractive forces between water and naphthalene molecules are $\boldsymbol{\text{weaker}}$ than the attractive forces between the water molecules themselves.”
- “In the naphthalene solution, water molecules are more likely to $\boldsymbol{\text{stay with other water molecules}}$ (or “exclude naphthalene,” “remain clustered,” etc.).”
Final Answers (Filling the Dropdowns):
- Lack of charged regions on: $\boldsymbol{\text{naphthalene}}$
- Attractive forces: $\boldsymbol{\text{weaker}}$
- Water molecules are more likely to: $\boldsymbol{\text{stay with other water molecules}}$ (or similar phrasing about preferring water-water interactions)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze the properties of naphthalene and methyl acetate:
Step 1: Recall Solubility Principles
Water is polar ($\ce{H2O}$ has a bent structure, polar bonds, and a net dipole). Polar substances or those with charged regions (like $\ce{-OH}$, $\ce{-COOH}$, or ionic groups) dissolve in water (like dissolves like). Nonpolar substances (no net dipole, no charged regions) do not dissolve well in water.
Step 2: Analyze Naphthalene
Naphthalene ($\ce{C10H8}$) is a nonpolar molecule (consists of only carbon and hydrogen, with a symmetrical aromatic structure). It has no charged regions (no polar bonds with large electronegativity differences, no ions). Thus, it cannot interact favorably with polar water molecules.
Step 3: Analyze Methyl Acetate
Methyl acetate ($\ce{CH3COOCH3}$) has a polar carbonyl group ($\ce{C=O}$) and polar $\ce{C-O}$ bonds. It has charged regions (partial positive on $\ce{C}$ of $\ce{C=O}$, partial negative on $\ce{O}$ of $\ce{C=O}$) that can interact with water’s polar regions.
Step 4: Solubility Difference
The difference in solubility arises because naphthalene lacks charged regions. Attractive forces between water and naphthalene are weaker than water-water forces (since naphthalene is nonpolar). In a naphthalene “solution” (actually insoluble), water molecules prefer to stay with other water molecules (so naphthalene is insoluble, and water molecules “stay together” or “exclude” naphthalene).
Filling the Blanks:
- “The difference in solubility between the two substances is due to the lack of charged regions on $\boldsymbol{\text{naphthalene}}$.”
- “Because of this, the attractive forces between water and naphthalene molecules are $\boldsymbol{\text{weaker}}$ than the attractive forces between the water molecules themselves.”
- “In the naphthalene solution, water molecules are more likely to $\boldsymbol{\text{stay with other water molecules}}$ (or “exclude naphthalene,” “remain clustered,” etc.).”
Final Answers (Filling the Dropdowns):
- Lack of charged regions on: $\boldsymbol{\text{naphthalene}}$
- Attractive forces: $\boldsymbol{\text{weaker}}$
- Water molecules are more likely to: $\boldsymbol{\text{stay with other water molecules}}$ (or similar phrasing about preferring water-water interactions)