QUESTION IMAGE
Question
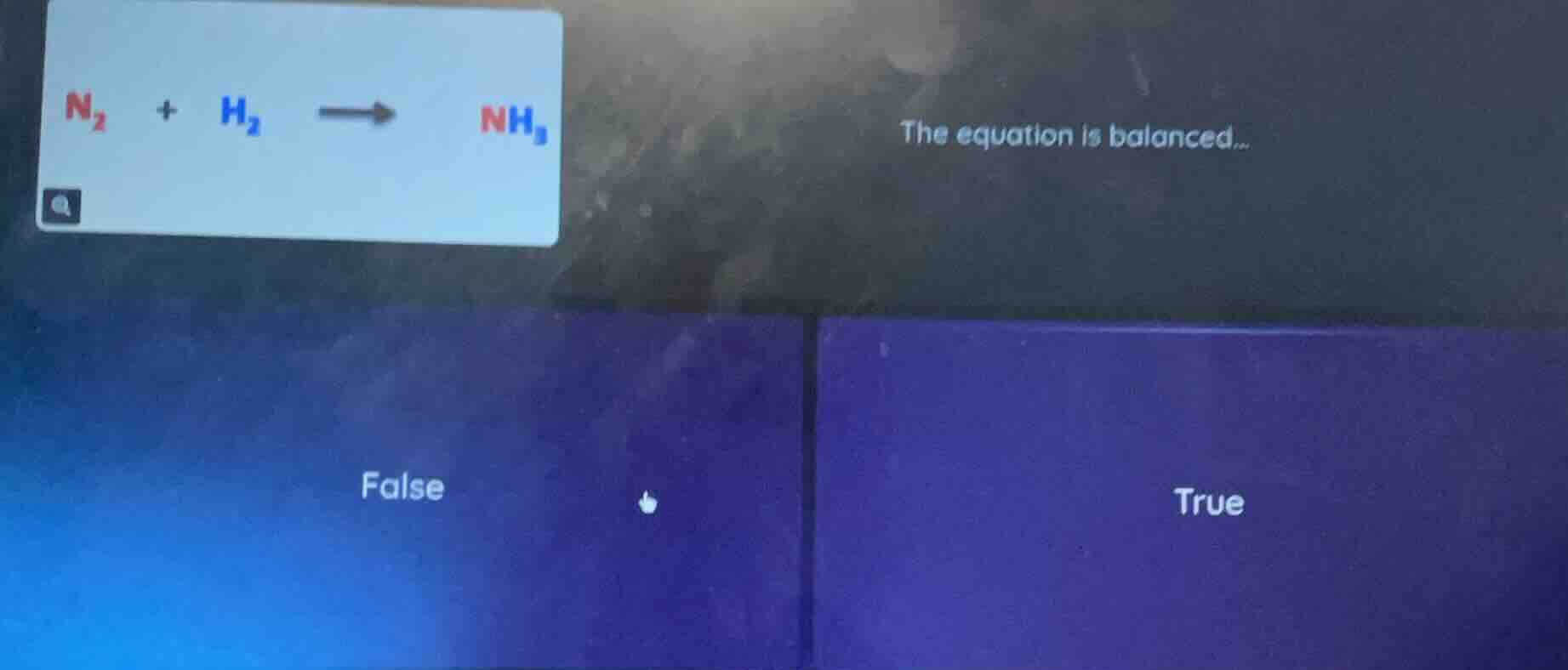
n₂ + h₂ → nh₃ the equation is balanced... false true
Step1: Count N atoms
Left side (reactants): \( \ce{N2} \) has 2 N atoms.
Right side (products): \( \ce{NH3} \) has 1 N atom (per molecule).
Step2: Count H atoms
Left side: \( \ce{H2} \) has 2 H atoms.
Right side: \( \ce{NH3} \) has 3 H atoms (per molecule).
Step3: Check balance
For N: 2 (left) vs. 1 (right) – not equal.
For H: 2 (left) vs. 3 (right) – not equal.
Thus, the equation is unbalanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
False