QUESTION IMAGE
Question
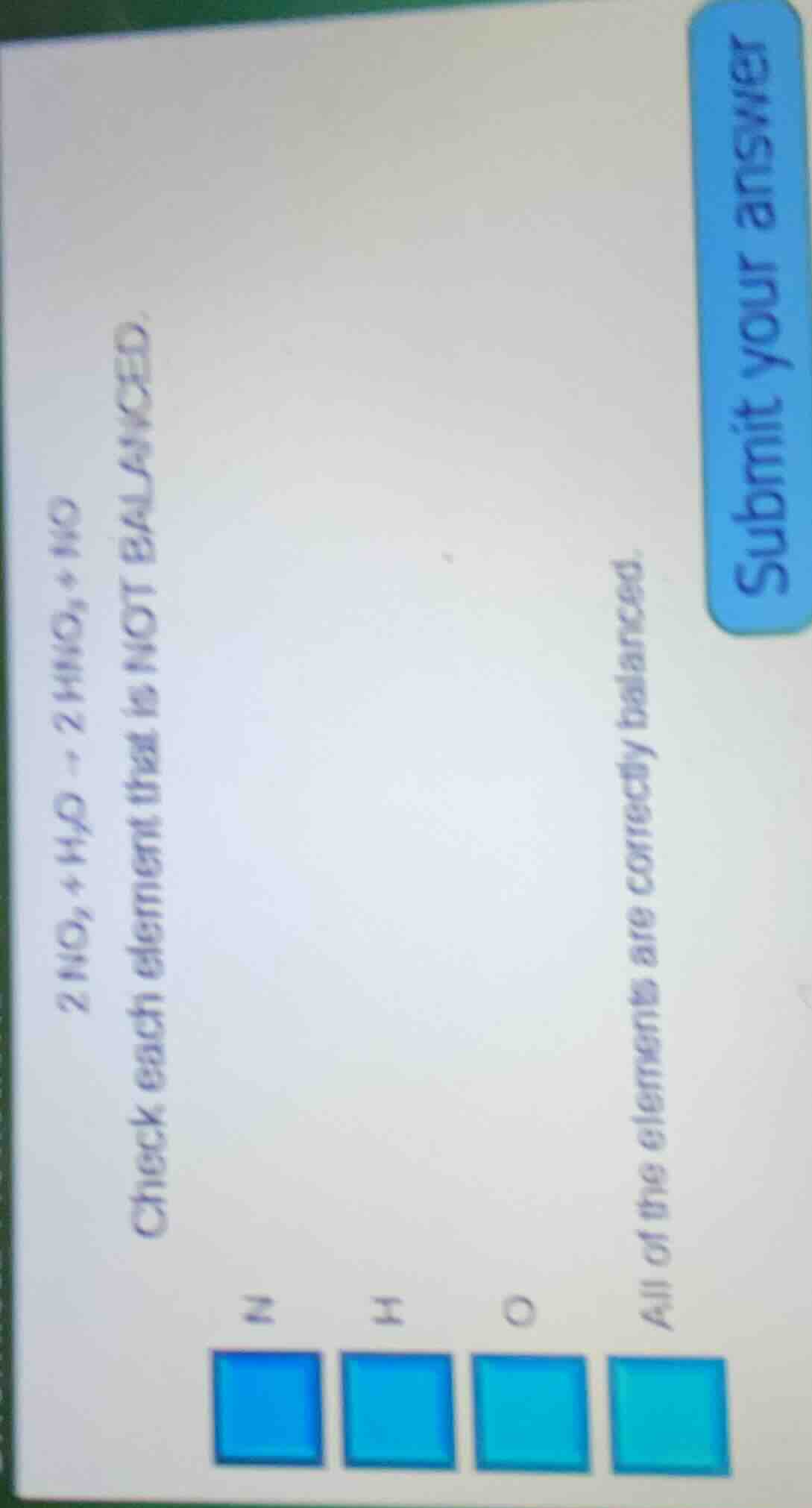
2 no₂ + h₂o → 2 hno₃ + no check each element that is not balanced. n h o all of the elements are correctly balanced. submit your answer
Step1: Count N atoms
Left side: In \(2\text{HNO}_3\), N atoms: \(2\times1 = 2\); In \(2\text{NO}_2\), N atoms: \(2\times1 = 2\). Total left N: \(2 + 2=4\)? Wait, no, the reaction is \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\)? Wait, original reaction: \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\)? Wait, user's reaction: \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\)? Wait, let's re - check.
Wait, given reaction: \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\) (assuming the given is \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\)).
Count N:
Left: \(2\text{NO}_2\) has \(2\) N, \(\text{H}_2\text{O}\) has \(0\) N. Total N left: \(2\).
Right: \(2\text{HNO}_3\) has \(2\times1 = 2\) N, \(\text{NO}\) has \(1\) N. Total N right: \(2 + 1=3\). So N is unbalanced? Wait, maybe the reaction is \(3\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\). Wait, user's reaction: \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\). Let's count each element:
For N:
Left: \(2\text{NO}_2\): \(2\) N atoms.
Right: \(2\text{HNO}_3\): \(2\) N atoms, \(\text{NO}\): \(1\) N atom. Total N right: \(2 + 1=3\). So N: left \(2\), right \(3\) → unbalanced? Wait, maybe I misread the reaction. Wait, the user's reaction is \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\)? Let's check H:
For H:
Left: \(\text{H}_2\text{O}\) has \(2\) H.
Right: \(2\text{HNO}_3\) has \(2\times1 = 2\) H. So H is balanced (2 = 2).
For O:
Left: \(2\text{NO}_2\): \(2\times2 = 4\) O; \(\text{H}_2\text{O}\): \(1\) O. Total O left: \(4 + 1=5\).
Right: \(2\text{HNO}_3\): \(2\times3 = 6\) O; \(\text{NO}\): \(1\) O. Total O right: \(6+1 = 7\)? No, that can't be. Wait, maybe the reaction is \(3\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\). Let's recalculate with \(3\text{NO}_2\):
N: Left \(3\), Right \(2 + 1=3\) → balanced.
H: Left \(2\), Right \(2\) → balanced.
O: Left \(3\times2+1 = 7\); Right \(2\times3 + 1=7\) → balanced.
But the user's reaction is \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\). Let's recalculate:
N: Left \(2\), Right \(2 + 1=3\) → unbalanced.
H: Left \(2\), Right \(2\) → balanced.
O: Left \(2\times2+1 = 5\); Right \(2\times3+1 = 7\) → unbalanced.
Wait, maybe the reaction is written as \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\) (user's given). Let's check each element:
- N:
- Left: \(2\text{NO}_2\) has \(2\) N atoms.
- Right: \(2\text{HNO}_3\) has \(2\) N, \(\text{NO}\) has \(1\) N. Total \(3\) N. So N is unbalanced.
- H:
- Left: \(\text{H}_2\text{O}\) has \(2\) H.
- Right: \(2\text{HNO}_3\) has \(2\) H. So H is balanced.
- O:
- Left: \(2\text{NO}_2\) (4 O)+\(\text{H}_2\text{O}\) (1 O) = 5 O.
- Right: \(2\text{HNO}_3\) (6 O)+\(\text{NO}\) (1 O) = 7 O. So O is unbalanced. Wait, but the question is to check which element is not balanced. Wait, maybe I made a mistake. Let's re - express the reaction:
Given reaction: \(2\text{NO}_2+\text{H}_2\text{O}
ightarrow2\text{HNO}_3+\text{NO}\)
Count N:
Left: \(2\) (from \(2\text{NO}_2\))
Right: \(2\) (from \(2\text{HNO}_3\))+\(1\) (from \(\text{NO}\))=\(3\). So N: 2 vs 3 → unbalanced.
Count H:
Left: \(2\) (from \(\text{H}_2\text{O}\))
Right: \(2\) (from \(2\text{HNO}_3\)) → balanced.
Count O:
Left: \(2\times2\) (from \(2\text{NO}_2\))+\(1\) (from \(\text{H}_2\text{O}\)) = \(5\)
Right: \(2\times3\) (from \(2\text{HNO}_3\))+\(1\) (from \(\text{NO}\)) = \(7\) → unbalanced.
But t…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
N