QUESTION IMAGE
Question
nomenclature of covalent compounds
quiz complete
1 2 3 4 5 6 7 8 9 10
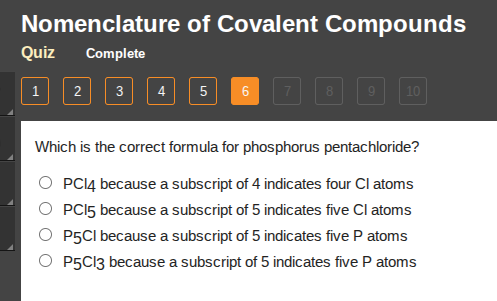
which is the correct formula for phosphorus pentachloride?
○ pcl4 because a subscript of 4 indicates four cl atoms
○ pcl5 because a subscript of 5 indicates five cl atoms
○ p5cl because a subscript of 5 indicates five p atoms
○ p5cl3 because a subscript of 5 indicates five p atoms
P, "chloride" = Cl.
Step2: Match name to formula
Phosphorus pentachloride means 1 P atom and 5 Cl atoms, so formula is $\text{PCl}_5$.
Step3: Evaluate options
Only the second option correctly links the prefix "penta-" to 5 Cl atoms in $\text{PCl}_5$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
P, "chloride" = Cl.
Step2: Match name to formula
Phosphorus pentachloride means 1 P atom and 5 Cl atoms, so formula is $\text{PCl}_5$.
Step3: Evaluate options
Only the second option correctly links the prefix "penta-" to 5 Cl atoms in $\text{PCl}_5$.