QUESTION IMAGE
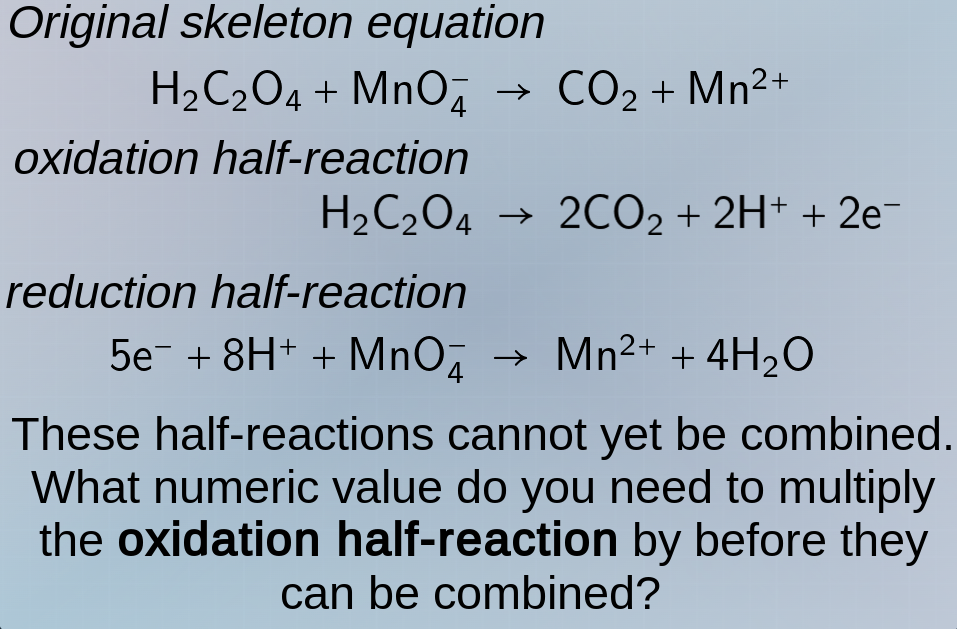
Question
original skeleton equation
h₂c₂o₄ + mno₄⁻ → co₂ + mn²⁺
oxidation half - reaction
h₂c₂o₄ → 2co₂ + 2h⁺ + 2e⁻
reduction half - reaction
5e⁻ + 8h⁺ + mno₄⁻ → mn²⁺ + 4h₂o
these half - reactions cannot yet be combined.
what numeric value do you need to multiply the oxidation half - reaction by before they can be combined?
Step1: Identify electrons in half-reactions
Oxidation half-reaction: \( \ce{H2C2O4 -> 2CO2 + 2H+ + 2e-} \) (loses 2 \( e^- \))
Reduction half-reaction: \( \ce{5e- + 8H+ + MnO4- -> Mn^2+ + 4H2O} \) (gains 5 \( e^- \))
Step2: Find least common multiple (LCM) of electrons
LCM of 2 and 5 is 10. To make oxidation half-reaction lose 10 \( e^- \), multiply by \( \frac{10}{2} = 5 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
5