QUESTION IMAGE
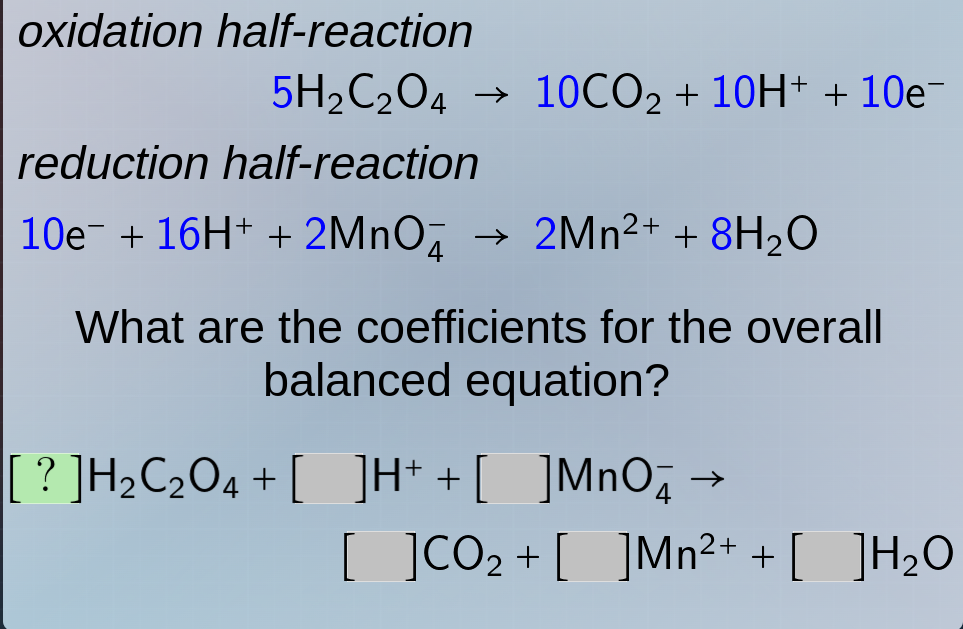
Question
oxidation half - reaction
$5\ce{h_{2}c_{2}o_{4}} \
ightarrow 10\ce{co_{2}} + 10\ce{h^{+}} + 10\ce{e^{-}}$
reduction half - reaction
$10\ce{e^{-}} + 16\ce{h^{+}} + 2\ce{mno^{-}_{4}} \
ightarrow 2\ce{mn^{2+}} + 8\ce{h_{2}o}$
what are the coefficients for the overall balanced equation?
$?\ce{h_{2}c_{2}o_{4}} + \quad\ce{h^{+}} + \quad\ce{mno^{-}_{4}} \
ightarrow \quad\ce{co_{2}} + \quad\ce{mn^{2+}} + \quad\ce{h_{2}o}$
Step1: Add the two half - reactions
The oxidation half - reaction is \(5H_{2}C_{2}O_{4}
ightarrow10CO_{2}+10H^{+}+10e^{-}\) and the reduction half - reaction is \(10e^{-}+16H^{+}+2MnO_{4}^{-}
ightarrow2Mn^{2+}+8H_{2}O\). When we add these two half - reactions together, the electrons (\(10e^{-}\)) will cancel out.
So, adding the left - hand sides: \(5H_{2}C_{2}O_{4}+10e^{-}+16H^{+}+2MnO_{4}^{-}\)
Adding the right - hand sides: \(10CO_{2}+10H^{+}+10e^{-}+2Mn^{2+}+8H_{2}O\)
After canceling the electrons, we can simplify the \(H^{+}\) terms. The number of \(H^{+}\) on the left is \(16\) and on the right is \(10\). So, the net \(H^{+}\) on the left after cancellation is \(16 - 10=6\).
Step2: Write the overall balanced equation
After adding and simplifying, the overall balanced equation is:
\(5H_{2}C_{2}O_{4}+6H^{+}+2MnO_{4}^{-}
ightarrow10CO_{2}+2Mn^{2+}+8H_{2}O\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The coefficients are \(5\) (for \(H_{2}C_{2}O_{4}\)), \(6\) (for \(H^{+}\)), \(2\) (for \(MnO_{4}^{-}\)), \(10\) (for \(CO_{2}\)), \(2\) (for \(Mn^{2+}\)) and \(8\) (for \(H_{2}O\)). So, filling in the blanks:
\(\boldsymbol{5}H_{2}C_{2}O_{4}+\boldsymbol{6}H^{+}+\boldsymbol{2}MnO_{4}^{-}
ightarrow\boldsymbol{10}CO_{2}+\boldsymbol{2}Mn^{2+}+\boldsymbol{8}H_{2}O\)