QUESTION IMAGE
Question
oxidation numbers
name ________ hour
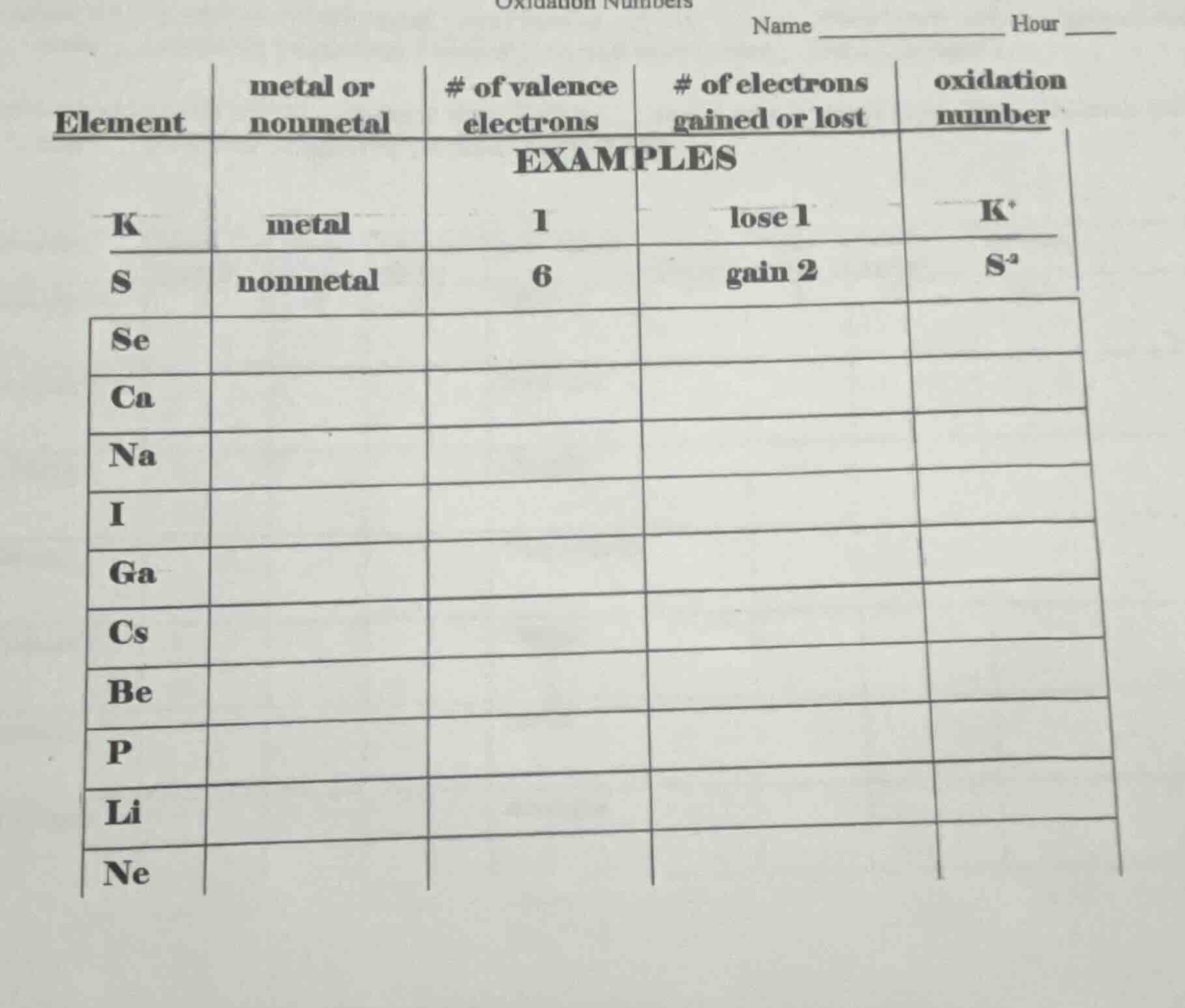
| element | metal or nonmetal | # of valence electrons | # of electrons gained or lost | oxidation number |
|---|---|---|---|---|
| k | metal | 1 | lose 1 | k⁺ |
| s | nonmetal | 6 | gain 2 | s²⁻ |
| se | ||||
| ca | ||||
| na | ||||
| i | ||||
| ga | ||||
| cs | ||||
| be | ||||
| p | ||||
| li | ||||
| ne |
To solve for each element, we analyze their properties (metal/nonmetal, valence electrons, electron gain/loss, oxidation number) using periodic table trends:
1. Selenium (Se)
Step1: Classify (Nonmetal, Group 16)
Nonmetal (Group 16, like S).
Step2: Valence Electrons
Group 16 → 6 valence electrons.
Step3: Electrons Gained/Lost
Nonmetals gain electrons to reach 8 (octet). Needs \( 8 - 6 = 2 \) electrons → gain 2.
Step4: Oxidation Number
Gains 2 → charge \( -2 \) → \( \text{Se}^{2-} \).
2. Calcium (Ca)
Step1: Classify (Metal, Group 2)
Metal (Group 2, alkaline earth metal).
Step2: Valence Electrons
Group 2 → 2 valence electrons.
Step3: Electrons Gained/Lost
Metals lose electrons. Loses 2 to reach octet → lose 2.
Step4: Oxidation Number
Loses 2 → charge \( +2 \) → \( \text{Ca}^{2+} \).
3. Sodium (Na)
Step1: Classify (Metal, Group 1)
Metal (Group 1, alkali metal).
Step2: Valence Electrons
Group 1 → 1 valence electron.
Step3: Electrons Gained/Lost
Loses 1 to reach octet → lose 1.
Step4: Oxidation Number
Loses 1 → charge \( +1 \) → \( \text{Na}^+ \).
4. Iodine (I)
Step1: Classify (Nonmetal, Group 17)
Nonmetal (Group 17, halogen).
Step2: Valence Electrons
Group 17 → 7 valence electrons.
Step3: Electrons Gained/Lost
Gains \( 8 - 7 = 1 \) electron → gain 1.
Step4: Oxidation Number
Gains 1 → charge \( -1 \) → \( \text{I}^- \).
5. Gallium (Ga)
Step1: Classify (Metal, Group 13)
Metal (Group 13, post - transition metal).
Step2: Valence Electrons
Group 13 → 3 valence electrons.
Step3: Electrons Gained/Lost
Loses 3 to reach octet → lose 3.
Step4: Oxidation Number
Loses 3 → charge \( +3 \) → \( \text{Ga}^{3+} \).
6. Cesium (Cs)
Step1: Classify (Metal, Group 1)
Metal (Group 1, alkali metal).
Step2: Valence Electrons
Group 1 → 1 valence electron.
Step3: Electrons Gained/Lost
Loses 1 → lose 1.
Step4: Oxidation Number
Loses 1 → charge \( +1 \) → \( \text{Cs}^+ \).
7. Beryllium (Be)
Step1: Classify (Metal, Group 2)
Metal (Group 2, alkaline earth metal).
Step2: Valence Electrons
Group 2 → 2 valence electrons.
Step3: Electrons Gained/Lost
Loses 2 → lose 2.
Step4: Oxidation Number
Loses 2 → charge \( +2 \) → \( \text{Be}^{2+} \).
8. Phosphorus (P)
Step1: Classify (Nonmetal, Group 15)
Nonmetal (Group 15).
Step2: Valence Electrons
Group 15 → 5 valence electrons.
Step3: Electrons Gained/Lost
Gains \( 8 - 5 = 3 \) electrons → gain 3.
Step4: Oxidation Number
Gains 3 → charge \( -3 \) → \( \text{P}^{3-} \).
9. Lithium (Li)
Step1: Classify (Metal, Group 1)
Metal (Group 1, alkali metal).
Step2: Valence Electrons
Group 1 → 1 valence electron.
Step3: Electrons Gained/Lost
Loses 1 → lose 1.
Step4: Oxidation Number
Loses 1 → charge \( +1 \) → \( \text{Li}^+ \).
10. Neon (Ne)
Step1: Classify (Nonmetal, Noble Gas)
Nonmetal (Group 18, noble gas).
Step2: Valence Electrons
Group 18 → 8 valence electrons (stable octet).
Step3: Electrons Gained/Lost
Noble gases do not gain/lose electrons (already stable) → 0.
Step4: Oxidation Number
No charge (stable) → 0 (or \( \text{Ne} \), no ion).
Filled Table (Key Columns):
| Element | Metal/Nonmetal | Valence Electrons | Electrons Gained/Lost | Oxidation Number |
|---|---|---|---|---|
| Ca | Metal | 2 | Lose 2 | \( \text{Ca}^{2+}… |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve for each element, we analyze their properties (metal/nonmetal, valence electrons, electron gain/loss, oxidation number) using periodic table trends:
1. Selenium (Se)
Step1: Classify (Nonmetal, Group 16)
Nonmetal (Group 16, like S).
Step2: Valence Electrons
Group 16 → 6 valence electrons.
Step3: Electrons Gained/Lost
Nonmetals gain electrons to reach 8 (octet). Needs \( 8 - 6 = 2 \) electrons → gain 2.
Step4: Oxidation Number
Gains 2 → charge \( -2 \) → \( \text{Se}^{2-} \).
2. Calcium (Ca)
Step1: Classify (Metal, Group 2)
Metal (Group 2, alkaline earth metal).
Step2: Valence Electrons
Group 2 → 2 valence electrons.
Step3: Electrons Gained/Lost
Metals lose electrons. Loses 2 to reach octet → lose 2.
Step4: Oxidation Number
Loses 2 → charge \( +2 \) → \( \text{Ca}^{2+} \).
3. Sodium (Na)
Step1: Classify (Metal, Group 1)
Metal (Group 1, alkali metal).
Step2: Valence Electrons
Group 1 → 1 valence electron.
Step3: Electrons Gained/Lost
Loses 1 to reach octet → lose 1.
Step4: Oxidation Number
Loses 1 → charge \( +1 \) → \( \text{Na}^+ \).
4. Iodine (I)
Step1: Classify (Nonmetal, Group 17)
Nonmetal (Group 17, halogen).
Step2: Valence Electrons
Group 17 → 7 valence electrons.
Step3: Electrons Gained/Lost
Gains \( 8 - 7 = 1 \) electron → gain 1.
Step4: Oxidation Number
Gains 1 → charge \( -1 \) → \( \text{I}^- \).
5. Gallium (Ga)
Step1: Classify (Metal, Group 13)
Metal (Group 13, post - transition metal).
Step2: Valence Electrons
Group 13 → 3 valence electrons.
Step3: Electrons Gained/Lost
Loses 3 to reach octet → lose 3.
Step4: Oxidation Number
Loses 3 → charge \( +3 \) → \( \text{Ga}^{3+} \).
6. Cesium (Cs)
Step1: Classify (Metal, Group 1)
Metal (Group 1, alkali metal).
Step2: Valence Electrons
Group 1 → 1 valence electron.
Step3: Electrons Gained/Lost
Loses 1 → lose 1.
Step4: Oxidation Number
Loses 1 → charge \( +1 \) → \( \text{Cs}^+ \).
7. Beryllium (Be)
Step1: Classify (Metal, Group 2)
Metal (Group 2, alkaline earth metal).
Step2: Valence Electrons
Group 2 → 2 valence electrons.
Step3: Electrons Gained/Lost
Loses 2 → lose 2.
Step4: Oxidation Number
Loses 2 → charge \( +2 \) → \( \text{Be}^{2+} \).
8. Phosphorus (P)
Step1: Classify (Nonmetal, Group 15)
Nonmetal (Group 15).
Step2: Valence Electrons
Group 15 → 5 valence electrons.
Step3: Electrons Gained/Lost
Gains \( 8 - 5 = 3 \) electrons → gain 3.
Step4: Oxidation Number
Gains 3 → charge \( -3 \) → \( \text{P}^{3-} \).
9. Lithium (Li)
Step1: Classify (Metal, Group 1)
Metal (Group 1, alkali metal).
Step2: Valence Electrons
Group 1 → 1 valence electron.
Step3: Electrons Gained/Lost
Loses 1 → lose 1.
Step4: Oxidation Number
Loses 1 → charge \( +1 \) → \( \text{Li}^+ \).
10. Neon (Ne)
Step1: Classify (Nonmetal, Noble Gas)
Nonmetal (Group 18, noble gas).
Step2: Valence Electrons
Group 18 → 8 valence electrons (stable octet).
Step3: Electrons Gained/Lost
Noble gases do not gain/lose electrons (already stable) → 0.
Step4: Oxidation Number
No charge (stable) → 0 (or \( \text{Ne} \), no ion).
Filled Table (Key Columns):
| Element | Metal/Nonmetal | Valence Electrons | Electrons Gained/Lost | Oxidation Number |
|---|---|---|---|---|
| Ca | Metal | 2 | Lose 2 | \( \text{Ca}^{2+} \) |
| Na | Metal | 1 | Lose 1 | \( \text{Na}^+ \) |
| I | Nonmetal | 7 | Gain 1 | \( \text{I}^- \) |
| Ga | Metal | 3 | Lose 3 | \( \text{Ga}^{3+} \) |
| Cs | Metal | 1 | Lose 1 | \( \text{Cs}^+ \) |
| Be | Metal | 2 | Lose 2 | \( \text{Be}^{2+} \) |
| P | Nonmetal | 5 | Gain 3 | \( \text{P}^{3-} \) |
| Li | Metal | 1 | Lose 1 | \( \text{Li}^+ \) |
| Ne | Nonmetal | 8 | 0 (no gain/loss) | 0 (or \( \text{Ne} \)) |
(Note: For Ne, oxidation number is 0 as it is inert and does not form ions under normal conditions.)