QUESTION IMAGE
Question
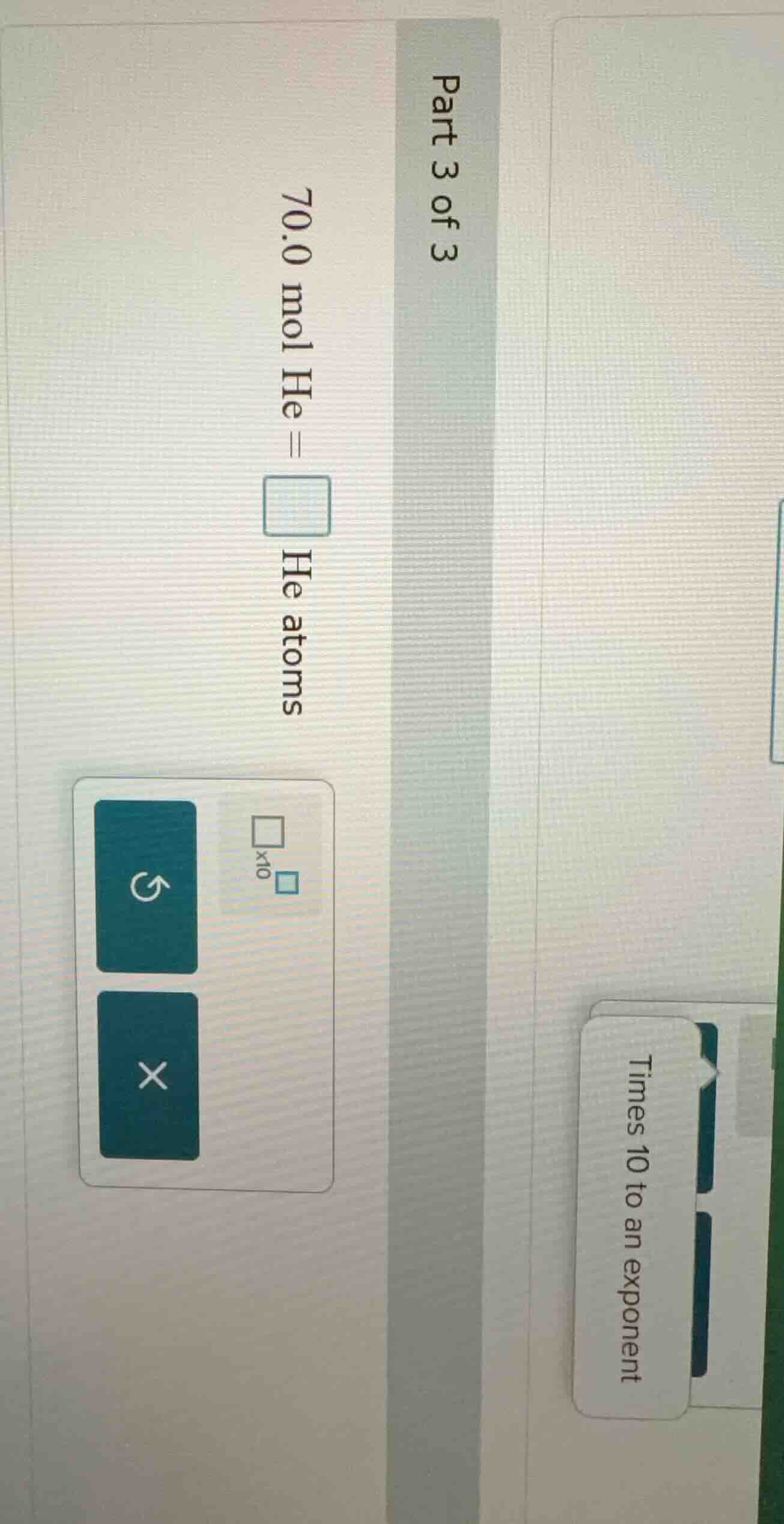
part 3 of 3
70.0 mol he =
he atoms
Step1: Recall Avogadro's number
Avogadro's number is \( 6.022 \times 10^{23} \) atoms/mol, which represents the number of atoms (or molecules) in one mole of a substance.
Step2: Calculate the number of He atoms
To find the number of He atoms in 70.0 mol of He, we multiply the number of moles by Avogadro's number. The formula is: Number of atoms = Moles × Avogadro's number.
So, we have \( 70.0 \, \text{mol} \times 6.022 \times 10^{23} \, \text{atoms/mol} \).
First, multiply 70.0 and 6.022: \( 70.0 \times 6.022 = 421.54 \).
Then, multiply by \( 10^{23} \): \( 421.54 \times 10^{23} = 4.2154 \times 10^{25} \) (after adjusting the scientific notation).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 4.22 \times 10^{25} \) (rounded to three significant figures)