QUESTION IMAGE
Question
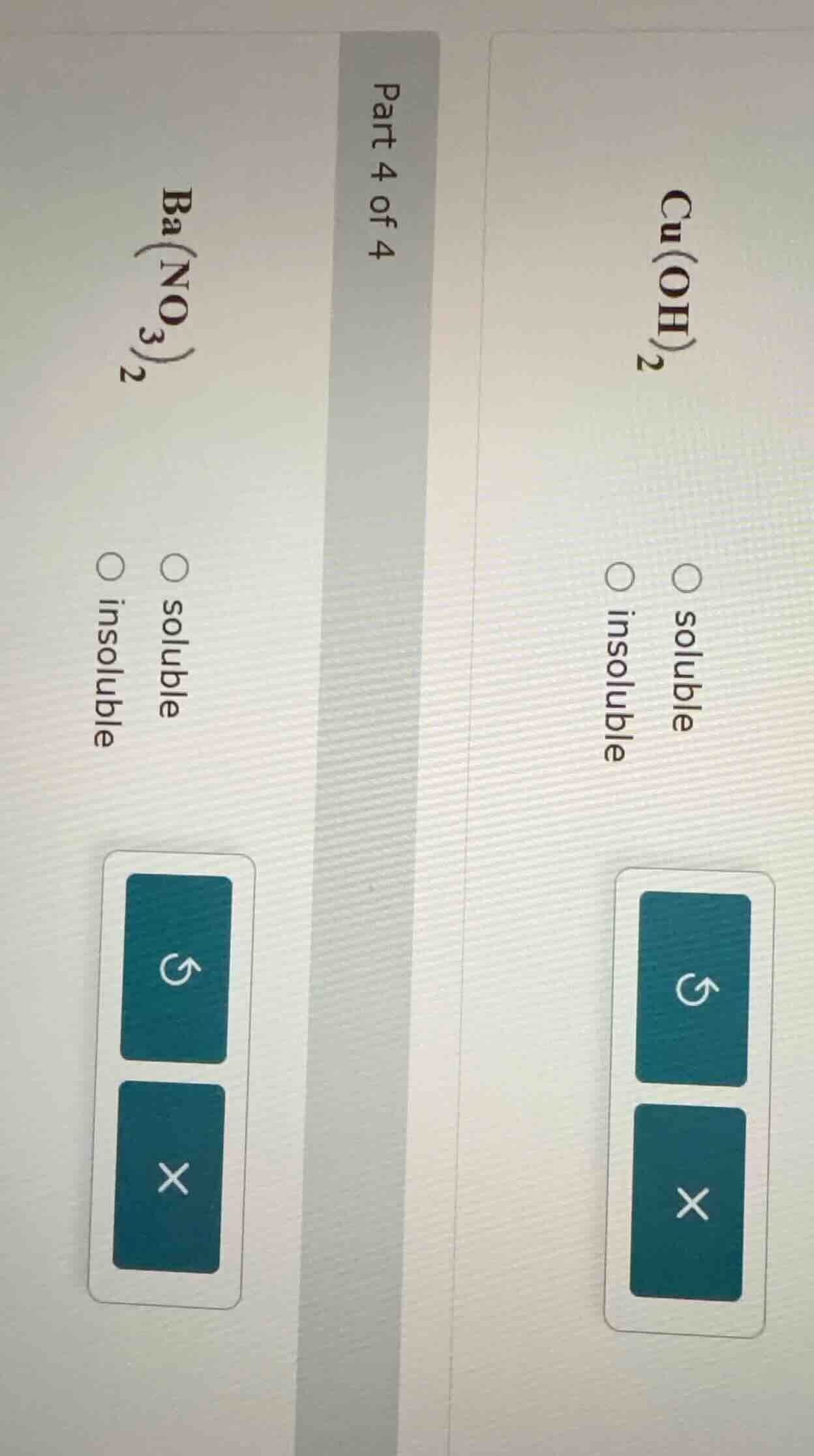
part 4 of 4
ba(no₃)₂
○ soluble
○ insoluble
cu(oh)₂
○ soluble
○ insoluble
Response
For $\boldsymbol{Cu(OH)_2}$:
Brief Explanations
Hydroxides of transition metals like copper are generally insoluble in water, following solubility rules.
Brief Explanations
Nitrates ($NO_3^-$) are almost always soluble, including barium nitrate, per solubility guidelines.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
insoluble