QUESTION IMAGE
Question
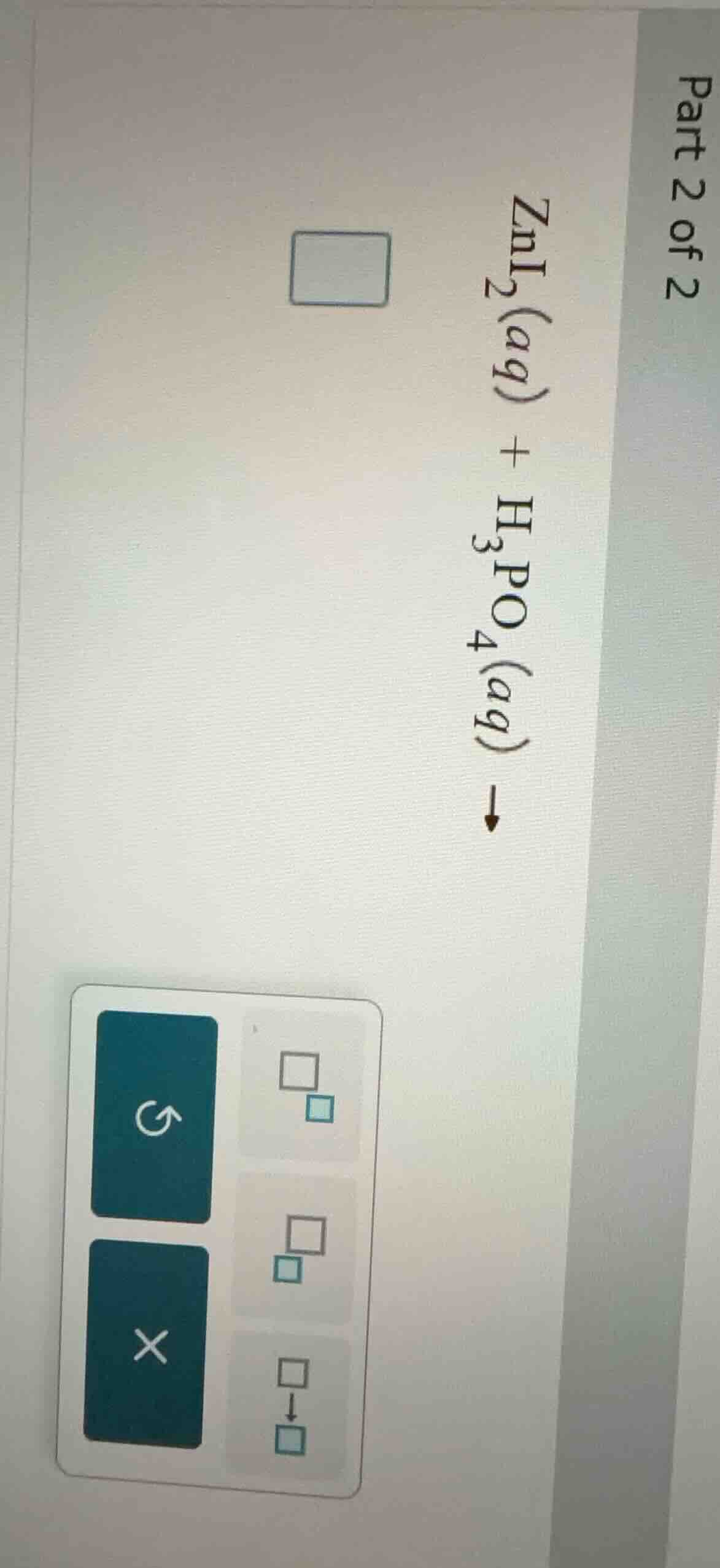
part 2 of 2
zni₂(aq) + h₃po₄(aq) →
Step 1: Identify the reaction type
This is a double - displacement reaction. In a double - displacement reaction, the general form is \(AB + CD
ightarrow AD+CB\). Here, \(AB = ZnI_2\) (\(A = Zn^{2 + }\), \(B=I^-\)) and \(CD = H_3PO_4\) (\(C = H^+\), \(D = PO_4^{3 - }\)).
Step 2: Determine the products
The cation of the first compound (\(Zn^{2+}\)) will combine with the anion of the second compound (\(PO_4^{3 - }\)) and the cation of the second compound (\(H^+\)) will combine with the anion of the first compound (\(I^-\)).
To balance the charges for the formation of \(Zn_3(PO_4)_2\): The charge of \(Zn\) is \(+ 2\) and the charge of \(PO_4^{3-}\) is \(-3\). Using the cross - multiplication method, the formula of the zinc phosphate is \(Zn_3(PO_4)_2\) (since \(3\times( + 2)+2\times(-3)=6 - 6 = 0\)).
For the formation of \(HI\): The charge of \(H^+\) is \(+ 1\) and the charge of \(I^-\) is \(-1\), so the formula is \(HI\).
Step 3: Balance the chemical equation
- Balance Zn atoms: There is 1 \(Zn\) in \(ZnI_2\) and 3 \(Zn\) in \(Zn_3(PO_4)_2\). So we put a coefficient of 3 in front of \(ZnI_2\): \(3ZnI_2(aq)+H_3PO_4(aq)
ightarrow Zn_3(PO_4)_2(s)+HI(aq)\)
- Balance \(PO_4\) groups: There is 1 \(PO_4\) group in \(H_3PO_4\) and 2 \(PO_4\) groups in \(Zn_3(PO_4)_2\). So we put a coefficient of 2 in front of \(H_3PO_4\): \(3ZnI_2(aq)+2H_3PO_4(aq)
ightarrow Zn_3(PO_4)_2(s)+HI(aq)\)
- Balance H and I atoms: There are 6 \(H\) and 6 \(I\) atoms from \(3ZnI_2\) and \(2H_3PO_4\) on the left - hand side. So we put a coefficient of 6 in front of \(HI\): \(3ZnI_2(aq)+2H_3PO_4(aq)
ightarrow Zn_3(PO_4)_2(s)+6HI(aq)\)
The balanced chemical equation is \(3ZnI_2(aq)+2H_3PO_4(aq)
ightarrow Zn_3(PO_4)_2(s)+6HI(aq)\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced equation is \(3ZnI_2(aq)+2H_3PO_4(aq)
ightarrow Zn_3(PO_4)_2(s)+6HI(aq)\) (If the question was to balance the equation. Since the original problem was a bit unclear but based on the reaction of \(ZnI_2\) and \(H_3PO_4\), this is the balanced chemical equation)