QUESTION IMAGE
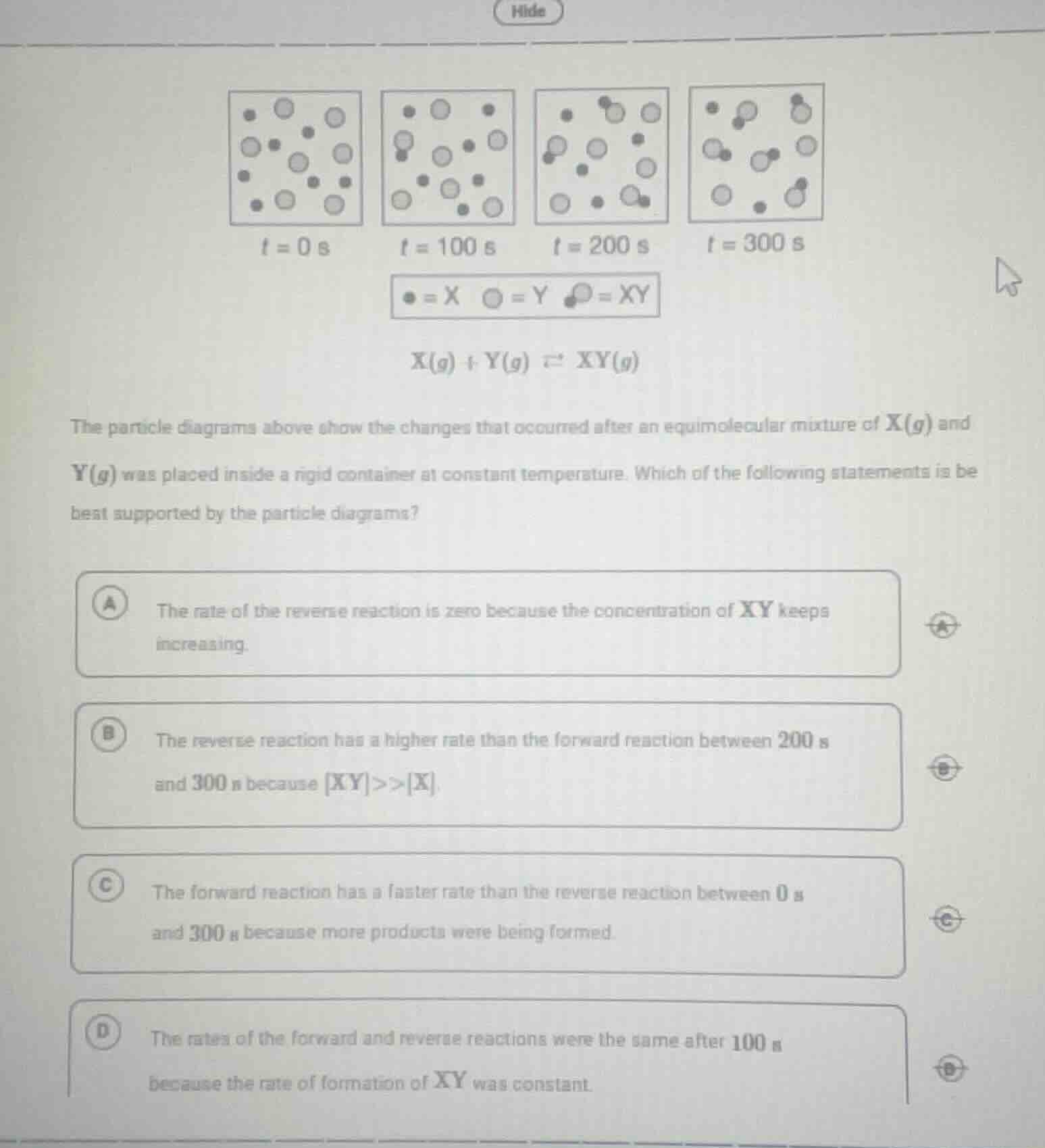
Question
the particle diagrams above show the changes that occurred after an equimolecular mixture of x(g) and y(g) was placed inside a rigid container at constant temperature. which of the following statements is best supported by the particle diagrams? a the rate of the reverse reaction is zero because the concentration of xy keeps increasing. b the reverse reaction has a higher rate than the forward reaction between 200 s and 300 s because xy >> x. c the forward reaction has a faster rate than the reverse reaction between 0 s and 300 s because more products were being formed. d the rates of the forward and reverse reactions were the same after 100 s because the rate of formation of xy was constant.
- Option A: In a reversible reaction, the reverse reaction rate is never zero as long as the product (XY) is present. As XY forms, the reverse reaction (XY decomposing to X and Y) starts occurring, so A is incorrect.
- Option B: Between 200 s and 300 s, we observe that the amount of XY is still increasing (more XY molecules are formed), which means the forward reaction rate is still higher than the reverse reaction rate. If the reverse reaction had a higher rate, the amount of XY would decrease. So B is incorrect.
- Option C: From t = 0 s to t = 300 s, the number of XY molecules is increasing (we can see more XY particles forming over time). This indicates that the forward reaction (X + Y → XY) is producing more products, so the forward reaction rate is faster than the reverse reaction rate (XY → X + Y) during this time interval. This statement is supported by the particle diagrams.
- Option D: After 100 s, we can see that the number of XY molecules is still changing (increasing) from 100 s to 200 s and 300 s. If the rates of forward and reverse reactions were the same, the concentrations (or number of particles) of reactants and products would be constant (equilibrium). Since the amount of XY is still increasing, the system is not at equilibrium at 100 s, so the rates are not the same. Thus, D is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. The forward reaction has a faster rate than the reverse reaction between 0 s and 300 s because more products were being formed.