QUESTION IMAGE
Question
period:
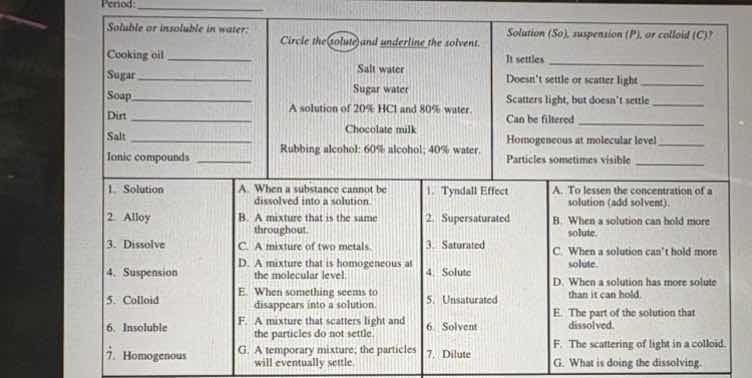
soluble or insoluble in water:
cooking oil
sugar
soap
dirt
salt
ionic compounds
circle the solute and underline the solvent.
salt water
sugar water
a solution of 20% hcl and 80% water.
chocolate milk
rubbing alcohol: 60% alcohol; 40% water.
solution (so), suspension (p), or colloid (c)?
it settles
doesnt settle or scatter light
scatters light, but doesnt settle
can be filtered
homogeneous at molecular level
particles sometimes visible
- solution
- alloy
- dissolve
- suspension
- colloid
- insoluble
- homogeneous
a. when a substance cannot be dissolved into a solution.
b. a mixture that is the same throughout.
c. a mixture of two metals.
d. a mixture that is homogeneous at the molecular level.
e. when a solute seems to disappear into a solution.
f. a mixture that scatters light and the particles do not settle.
g. a temporary mixture; the particles will eventually settle.
- tyndall effect
- supersaturated
- saturated
- solute
- unsaturated
- solvent
- dilute
a. to lessen the concentration of a solution (add solvent).
b. when a solution can hold more solute.
c. when a solution cant hold more solute.
d. when a solution has more solute than it can hold.
e. the part of the solution that dissolved.
f. the scattering of light in a colloid.
g. what is doing the dissolving.
Part 1: Soluble or Insoluble in water
Step1: Classify solubility in water
Cooking oil: Nonpolar, insoluble in polar water.
Sugar: Polar, dissolves in water.
Soap: Has polar/nonpolar ends, soluble.
Dirt: Heterogeneous, insoluble.
Salt: Ionic, dissolves in polar water.
Ionic compounds: Most are soluble in polar water.
Step1: Identify solute (minor component)
Solute = substance dissolved; solvent = dissolving medium.
Step2: Mark solute/solvent for each mixture
Salt water: Solute = Salt; Solvent = Water
Sugar water: Solute = Sugar; Solvent = Water
20% HCl/80% water: Solute = 20% HCl; Solvent = 80% water
Chocolate milk: Solute = Chocolate solids; Solvent = Milk
Rubbing alcohol (60% alcohol/40% water): Solute = 40% water; Solvent = 60% alcohol (higher % is solvent)
Step1: Match property to mixture type
Settles = Suspension; No settle/scatter = Solution; Scatters light/no settle = Colloid; Filterable = Solution; Homogeneous molecular = Solution; Visible particles = Colloid
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Cooking oil: Insoluble
Sugar: Soluble
Soap: Soluble
Dirt: Insoluble
Salt: Soluble
Ionic compounds: Soluble (most)
---