QUESTION IMAGE
Question
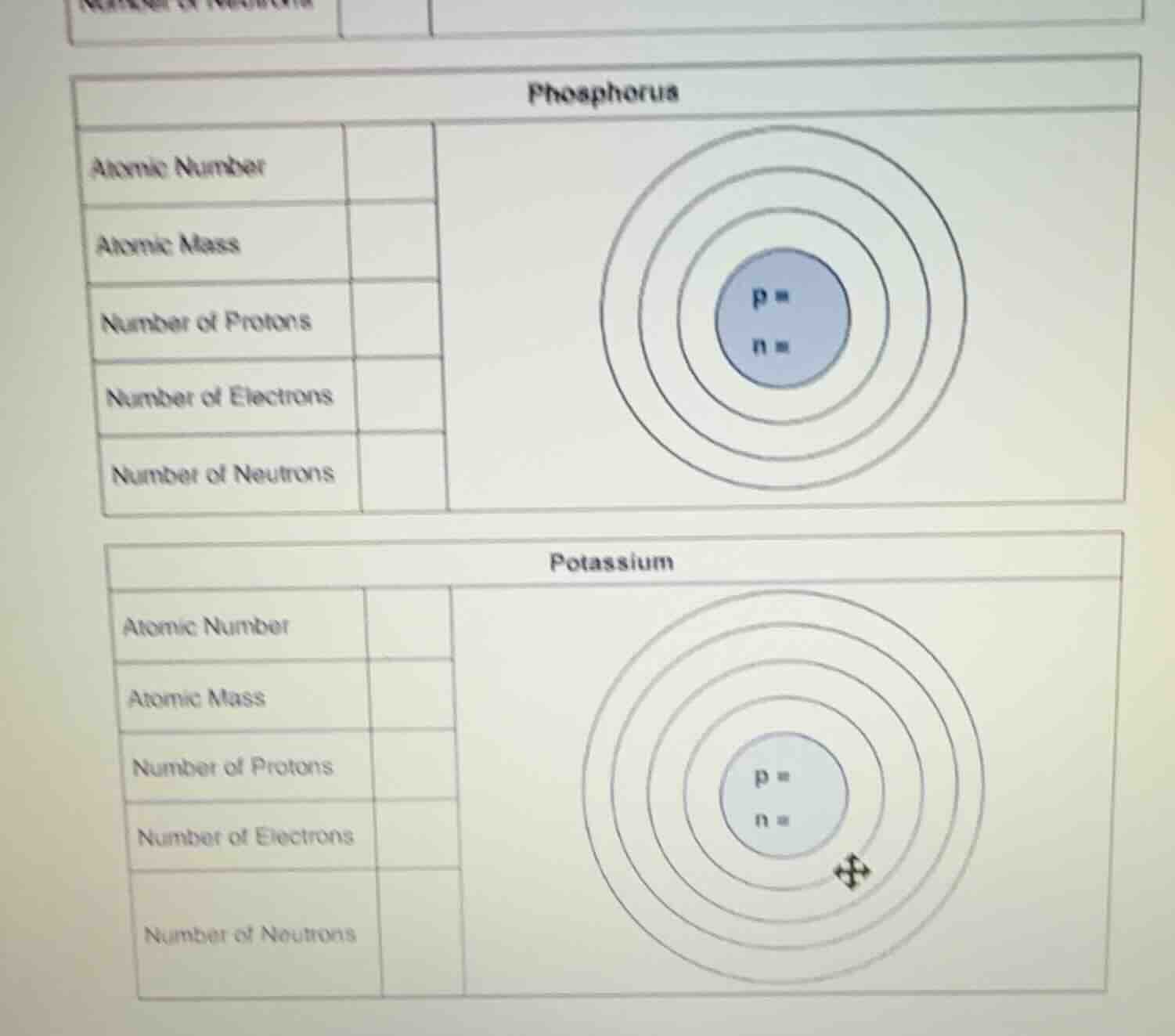
phosphorus
atomic number
atomic mass
number of protons
number of electrons
number of neutrons
p =
n =
potassium
atomic number
atomic mass
number of protons
number of electrons
number of neutrons
p =
n =
Step1: Get Phosphorus base values
From periodic table: Atomic number of Phosphorus (P) = 15, Atomic mass ≈ 31
Step2: Calculate P subatomic particles
Number of Protons = Atomic Number = $15$
Number of Electrons = Protons (neutral atom) = $15$
Number of Neutrons = Atomic Mass - Atomic Number = $31 - 15 = 16$
Step3: Get Potassium base values
From periodic table: Atomic number of Potassium (K) = 19, Atomic mass ≈ 39
Step4: Calculate K subatomic particles
Number of Protons = Atomic Number = $19$
Number of Electrons = Protons (neutral atom) = $19$
Number of Neutrons = Atomic Mass - Atomic Number = $39 - 19 = 20$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Phosphorus
| Category | Value |
|---|---|
| Atomic Mass | 31 |
| Number of Protons | 15 |
| Number of Electrons | 15 |
| Number of Neutrons | 16 |
(For the Bohr model: p = 15, n = 16; electrons: 2 in first shell, 8 in second, 5 in third)
Potassium
| Category | Value |
|---|---|
| Atomic Mass | 39 |
| Number of Protons | 19 |
| Number of Electrons | 19 |
| Number of Neutrons | 20 |
(For the Bohr model: p = 19, n = 20; electrons: 2 in first shell, 8 in second, 8 in third, 1 in fourth)