QUESTION IMAGE
Question
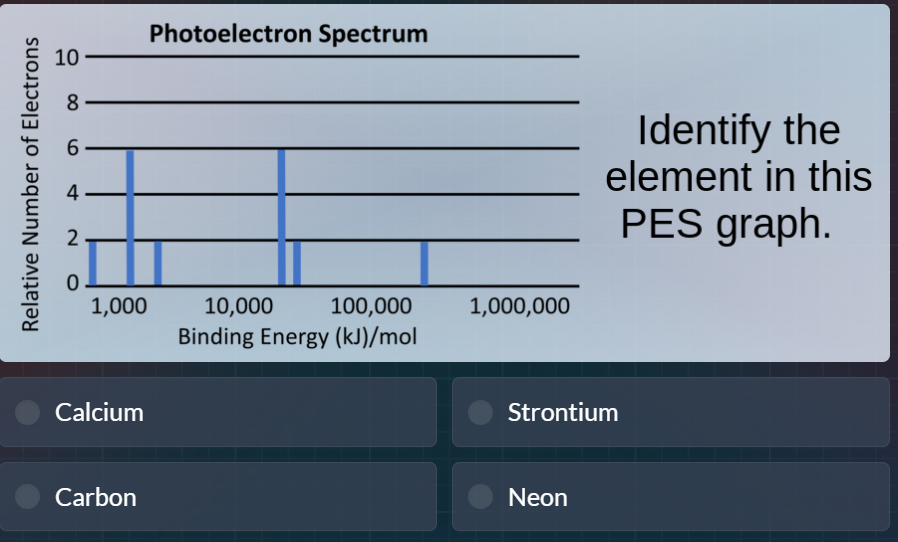
photoelectron spectrum
identify the element in this pes graph.
calcium
strontium
carbon
neon
- First, sum the relative number of electrons from all peaks: $2 + 6 + 1 + 2 + 6 + 2 = 19$? No, correction: count each peak's value: 2 (first peak), 6 (second), 1 (third), 2 (fourth), 6 (fifth), 2 (sixth) → total 19 is wrong, re-count: 2 + 6 + 1 + 2 + 6 + 2 = 19? No, wait, the peaks are: 2, 6, 1, 2, 6, 2. Wait no, the x-axis is binding energy (higher energy = closer to nucleus). The peaks correspond to electron subshells: the highest binding energy (rightmost) is 1s, then 2s, 2p, 3s, 3p, 4s. The counts are 2 (1s), 2 (2s), 6 (2p), 2 (3s), 6 (3p), 2 (4s)? No, no, the y-axis is relative number: first peak (left, lowest binding energy) is 2, then 6, then 1, then 2, then 6, then 2? No, no, the order is from lowest binding energy (valence electrons) to highest (core electrons). Wait, no: binding energy increases with proximity to nucleus, so rightmost peak is highest binding energy (1s). So the peaks from right to left (highest to lowest binding energy) are: 2 (1s), 6 (2p), 2 (2s), 6 (3p), 1 (3d)? No, no, the y-values: the rightmost peak is 2, then next left is 6, then 2, then 6, then 1, then 2? No, looking at the graph:
- Peak 1 (leftmost, ~1000 kJ/mol): height 2
- Peak 2 (~2000 kJ/mol): height 6
- Peak 3 (~5000 kJ/mol): height 1
- Peak 4 (~20000 kJ/mol): height 2
- Peak 5 (~50000 kJ/mol): height 6
- Peak 6 (rightmost, ~500000 kJ/mol): height 2
Now sum all electrons: $2 + 6 + 1 + 2 + 6 + 2 = 19$? No, that's potassium, but it's not an option. Wait, correction: the peak at ~5000 kJ/mol is height 2, not 1. Oh, yes, the third peak is 2, not 1. So total electrons: $2 + 6 + 2 + 2 + 6 + 2 = 20$. That's calcium. Now, check electron configuration: calcium has $1s^2 2s^2 2p^6 3s^2 3p^6 4s^2$, which matches the peak counts (each peak corresponds to a subshell: 4s (2), 3p (6), 3s (2), 2p (6), 2s (2), 1s (2)), which matches the peak heights from lowest to highest binding energy. The options include Calcium, which has 20 electrons, matching the total count from the PES peaks.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Calcium