QUESTION IMAGE
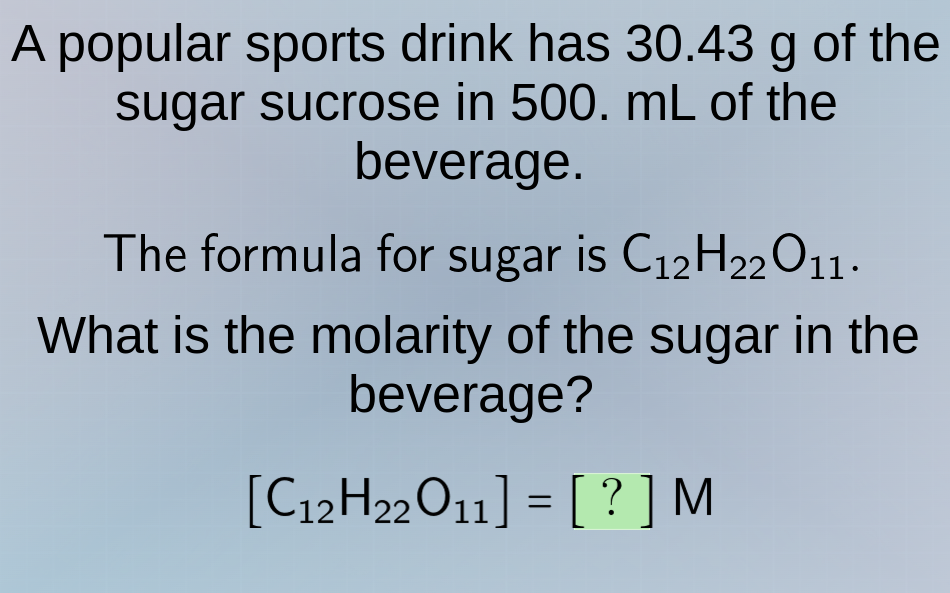
Question
a popular sports drink has 30.43 g of the sugar sucrose in 500. ml of the beverage. the formula for sugar is c₁₂h₂₂o₁₁. what is the molarity of the sugar in the beverage? c₁₂h₂₂o₁₁ = ? m
Step1: Calculate molar mass of sucrose
Molar mass of \( C_{12}H_{22}O_{11} \): \( 12\times12.01 + 22\times1.008 + 11\times16.00 \)
\( = 144.12 + 22.176 + 176.00 = 342.296 \, \text{g/mol} \)
Step2: Find moles of sucrose
Moles \( = \frac{\text{mass}}{\text{molar mass}} = \frac{30.43 \, \text{g}}{342.296 \, \text{g/mol}} \approx 0.0889 \, \text{mol} \)
Step3: Convert volume to liters
\( 500. \, \text{mL} = 0.500 \, \text{L} \)
Step4: Calculate molarity
Molarity \( = \frac{\text{moles}}{\text{volume (L)}} = \frac{0.0889 \, \text{mol}}{0.500 \, \text{L}} \approx 0.178 \, \text{M} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 0.178 \)