QUESTION IMAGE
Question
practice: isotope calculations #1
part 1 use the notation below to answer the following questions.
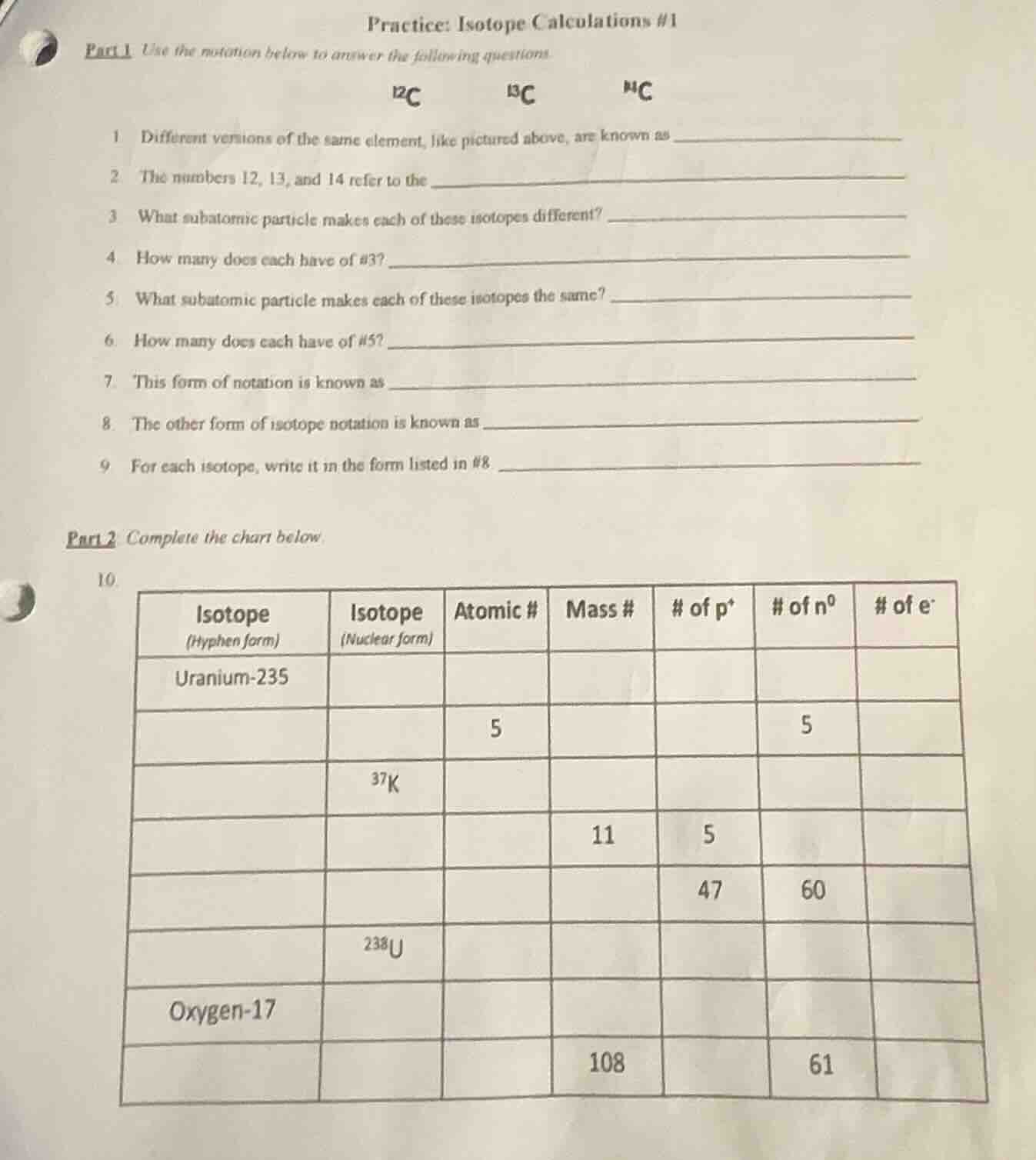
¹²c ¹³c ¹⁴c
1 different versions of the same element, like pictured above, are known as
2 the numbers 12, 13, and 14 refer to the
3 what subatomic particle makes each of these isotopes different?
4 how many does each have of #3?
5 what subatomic particle makes each of these isotopes the same?
6 how many does each have of #5?
7 this form of notation is known as
8 the other form of isotope notation is known as
9 for each isotope, write it in the form listed in #8
part 2 complete the chart below.
10.
| isotope (hyphen form) | isotope (nuclear form) | atomic # | mass # | # of p⁺ | # of n⁰ | # of e⁻ |
|---|---|---|---|---|---|---|
| 5 | 5 | |||||
| ³⁷k | ||||||
| 11 | 5 | |||||
| 47 | 60 | |||||
| ²³⁸u | ||||||
| oxygen-17 | ||||||
| 108 | 61 |
Part 1
1.
Different versions of the same element with different mass numbers (due to different neutron counts) are called isotopes.
The numbers 12, 13, 14 in \(^{12}\text{C}\), \(^{13}\text{C}\), \(^{14}\text{C}\) represent the mass number, which is the sum of protons and neutrons in the nucleus.
Isotopes of an element have the same number of protons (atomic number) but different numbers of neutrons, so neutrons are the subatomic particle that differs.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
isotopes
2.