QUESTION IMAGE
Question
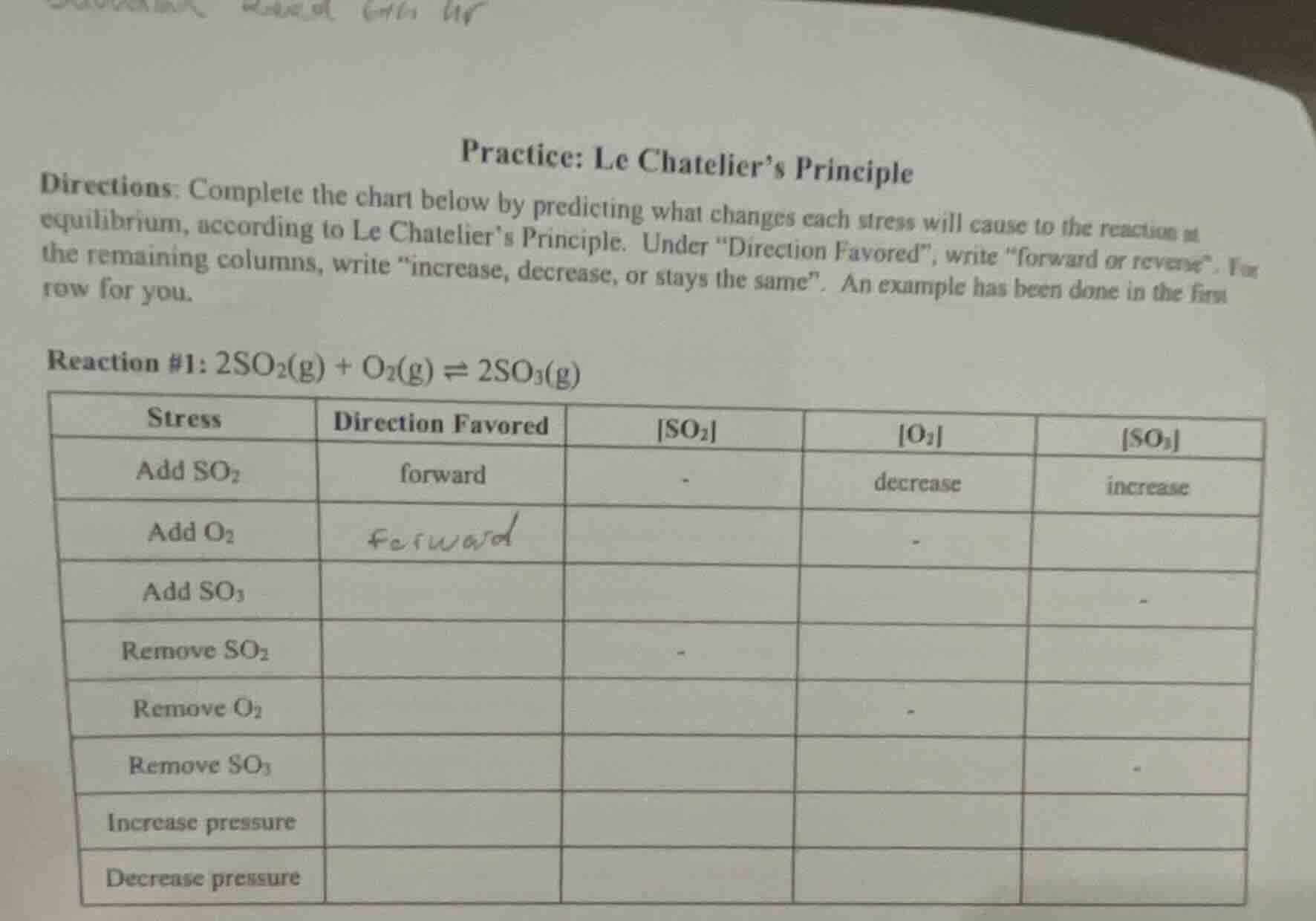
practice: le chateliers principle
directions: complete the chart below by predicting what changes each stress will cause to the reaction at equilibrium, according to le chateliers principle. under \direction favored\, write \forward or reverse\. for the remaining columns, write \increase, decrease, or stays the same\. an example has been done in the first row for you.
reaction #1: $2so_2(g) + o_2(g) \
ightleftharpoons 2so_3(g)$
| stress | direction favored | $so_2$ | $o_2$ | $so_3$ | ---- | ---- | ---- | ---- | ---- | add $so_2$ | forward | - | decrease | increase | add $o_2$ | forward | - | add $so_3$ | - | remove $so_2$ | - | remove $o_2$ | - | remove $so_3$ | - | increase pressure | decrease pressure |
Step1: Apply Le Chatelier's Principle (Add O₂)
Adding reactant favors forward reaction. $\ce{O2}$ is added (marked '-'), so $\ce{[SO2]}$ decreases, $\ce{[SO3]}$ increases.
Step2: Apply Le Chatelier's Principle (Add SO₃)
Adding product favors reverse reaction. $\ce{[SO3]}$ is added (marked '-'), so $\ce{[SO2]}$ increases, $\ce{[O2]}$ increases.
Step3: Apply Le Chatelier's Principle (Remove SO₂)
Removing reactant favors reverse reaction. $\ce{[SO2]}$ is removed (marked '-'), so $\ce{[O2]}$ increases, $\ce{[SO3]}$ decreases.
Step4: Apply Le Chatelier's Principle (Remove O₂)
Removing reactant favors reverse reaction. $\ce{[O2]}$ is removed (marked '-'), so $\ce{[SO2]}$ increases, $\ce{[SO3]}$ decreases.
Step5: Apply Le Chatelier's Principle (Remove SO₃)
Removing product favors forward reaction. $\ce{[SO3]}$ is removed (marked '-'), so $\ce{[SO2]}$ decreases, $\ce{[O2]}$ decreases.
Step6: Apply Le Chatelier's Principle (Increase pressure)
Fewer gas moles (3 reactant → 2 product) favor forward reaction. $\ce{[SO2]}$ decreases, $\ce{[O2]}$ decreases, $\ce{[SO3]}$ increases.
Step7: Apply Le Chatelier's Principle (Decrease pressure)
More gas moles favor reverse reaction. $\ce{[SO2]}$ increases, $\ce{[O2]}$ increases, $\ce{[SO3]}$ decreases.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Stress | Direction Favored | [SO₂] | [O₂] | [SO₃] |
|---|---|---|---|---|
| Add O₂ | forward | decrease | - | increase |
| Add SO₃ | reverse | increase | increase | - |
| Remove SO₂ | reverse | - | increase | decrease |
| Remove O₂ | reverse | increase | - | decrease |
| Remove SO₃ | forward | decrease | decrease | - |
| Increase pressure | forward | decrease | decrease | increase |
| Decrease pressure | reverse | increase | increase | decrease |