QUESTION IMAGE
Question
pre-lab assignment
answer these questions before you come to lab. show your work fully and clearly.
- a sample was prepared by mixing 15.0 ml of $6.00 \times 10^{-5}$ m crystal violet (cv) with 5.00 ml of 0.250 m naoh. calculate the resulting concentrations of cv and of $oh^-$.
- suppose that trial i in this lab was run using 0.10 m naoh, producing a linear kinetics graph with best-fit equation $y = -0.003008 + 0.785$. if trial ii was run using 0.30 m naoh, what would the slope of the same graph be if the reaction were (a) zero order, (b) first order, or (c) second order?
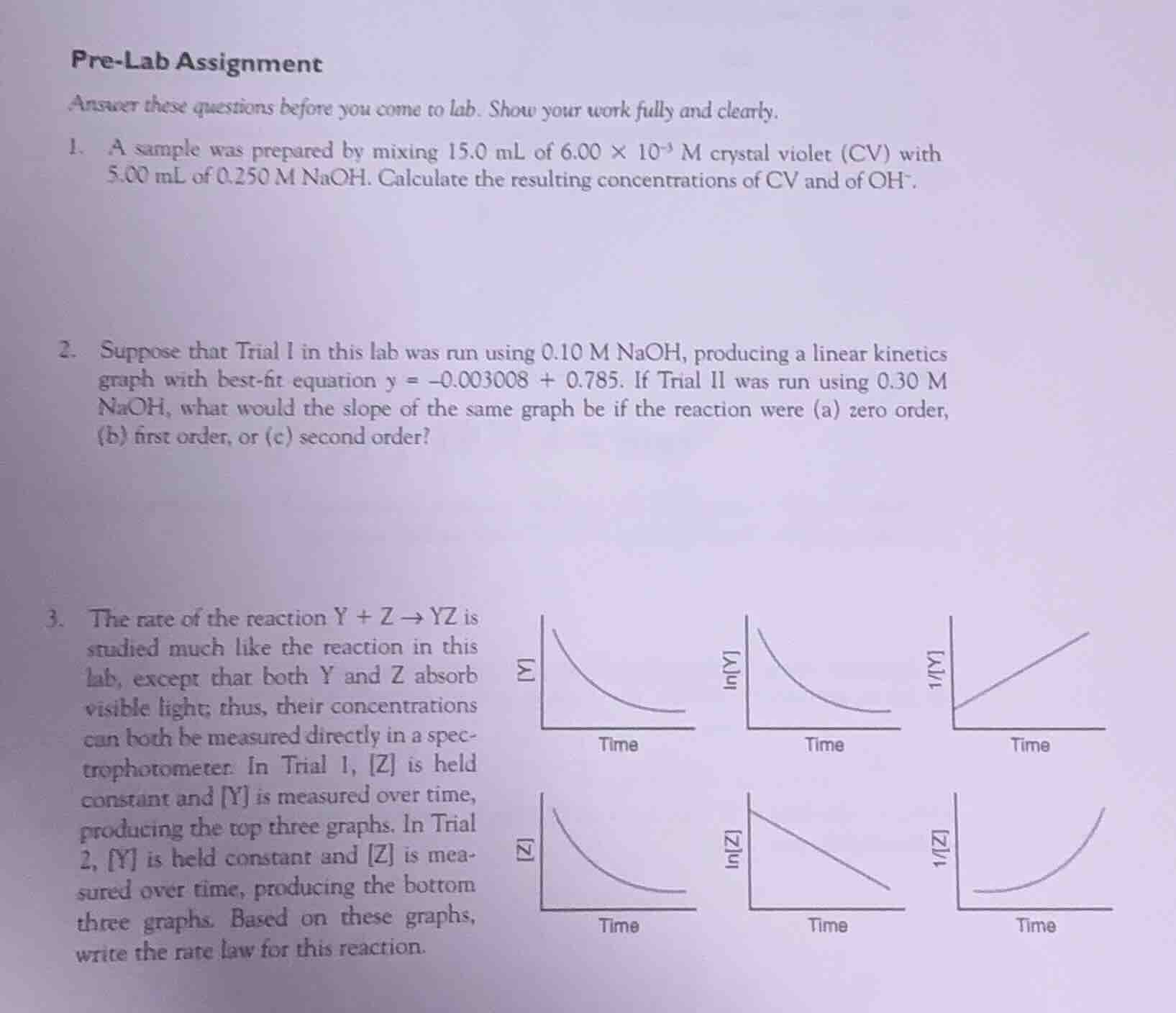
- the rate of the reaction $y + z \
ightarrow yz$ is studied much like the reaction in this lab, except that both y and z absorb visible light; thus, their concentrations can both be measured directly in a spectrophotometer. in trial 1, z is held constant and y is measured over time, producing the top three graphs. in trial 2, y is held constant and z is measured over time, producing the bottom three graphs. based on these graphs, write the rate law for this reaction.
Step1: Calculate total solution volume
$V_{\text{total}} = 15.0\ \text{mL} + 5.00\ \text{mL} = 20.0\ \text{mL}$
Step2: Find diluted CV concentration
Use $M_1V_1 = M_2V_2$:
$M_{CV,2} = \frac{M_{CV,1}V_{CV,1}}{V_{\text{total}}} = \frac{6.00 \times 10^{-5}\ \text{M} \times 15.0\ \text{mL}}{20.0\ \text{mL}}$
$M_{CV,2} = 4.50 \times 10^{-5}\ \text{M}$
Step3: Find diluted $\text{OH}^-$ concentration
Use $M_1V_1 = M_2V_2$:
$M_{\text{OH}^-,2} = \frac{M_{\text{NaOH},1}V_{\text{NaOH},1}}{V_{\text{total}}} = \frac{0.250\ \text{M} \times 5.00\ \text{mL}}{20.0\ \text{mL}}$
$M_{\text{OH}^-,2} = 0.0625\ \text{M}$
---
Step1: Define rate law for zero order
Rate $= k[\text{OH}^-]^0 = k$, slope $=-k$
Step2: Zero order slope (no $[\text{OH}^-]$ dependence)
$\text{Slope} = -0.003008$
Step3: Define rate law for first order
Rate $= k[\text{OH}^-]^1$, slope $=-k[\text{OH}^-]$
Step4: First order slope (3x $[\text{OH}^-]$)
$\text{Slope} = -0.003008 \times 3 = -0.009024$
Step5: Define rate law for second order
Rate $= k[\text{OH}^-]^2$, slope $=-k[\text{OH}^-]^2$
Step6: Second order slope (9x $[\text{OH}^-]^2$)
$\text{Slope} = -0.003008 \times 3^2 = -0.027072$
---
Step1: Identify order for reactant Y
Only $1/[\text{Y}]$ vs time is linear: order = 2
Step2: Identify order for reactant Z
Only $\ln[\text{Z}]$ vs time is linear: order = 1
Step3: Write combined rate law
Rate $= k[\text{Y}]^2[\text{Z}]^1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Concentration of CV: $\boldsymbol{4.50 \times 10^{-5}\ \text{M}}$; Concentration of $\text{OH}^-$: $\boldsymbol{0.0625\ \text{M}}$
- (a) Zero order: $\boldsymbol{-0.003008}$; (b) First order: $\boldsymbol{-0.009024}$; (c) Second order: $\boldsymbol{-0.027072}$
- $\boldsymbol{\text{Rate} = k[\text{Y}]^2[\text{Z}]}$