QUESTION IMAGE
Question
problem 9 of 56
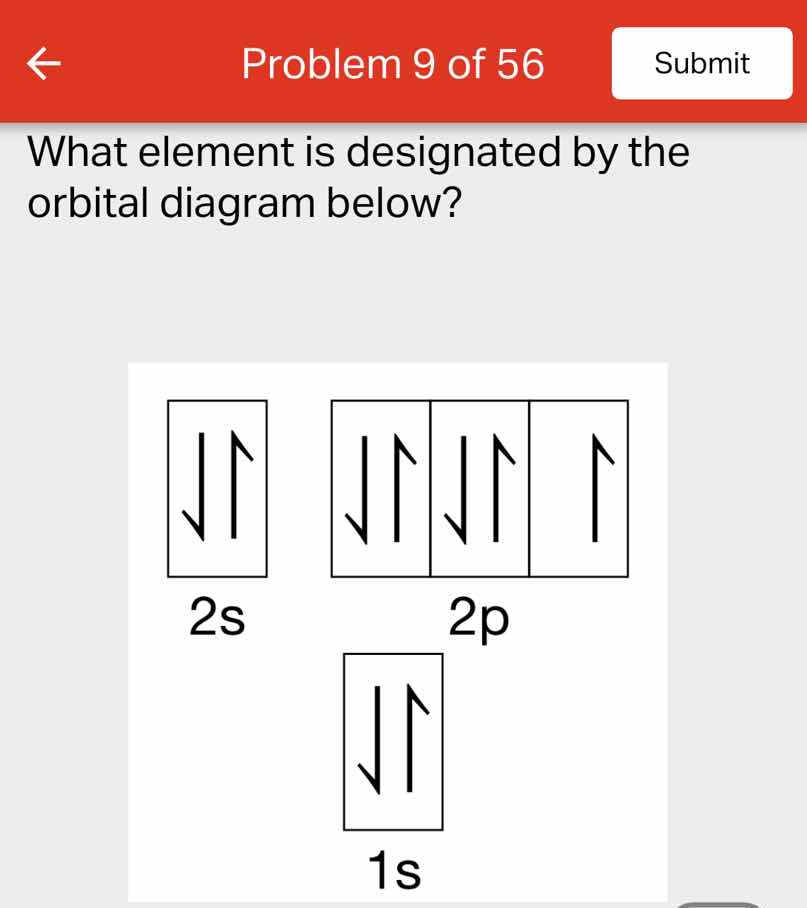
what element is designated by the orbital diagram below?
orbital diagram: 1s with paired electrons, 2s with paired electrons, 2p with two paired and one unpaired electron
Step1: Count total electrons
1s: 2, 2s: 2, 2p: 3. Total = $2+2+3=7$
Step2: Match electrons to element
Neutral element has atomic number = electron count. Atomic number 7 is nitrogen.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Nitrogen (N)