QUESTION IMAGE
Question
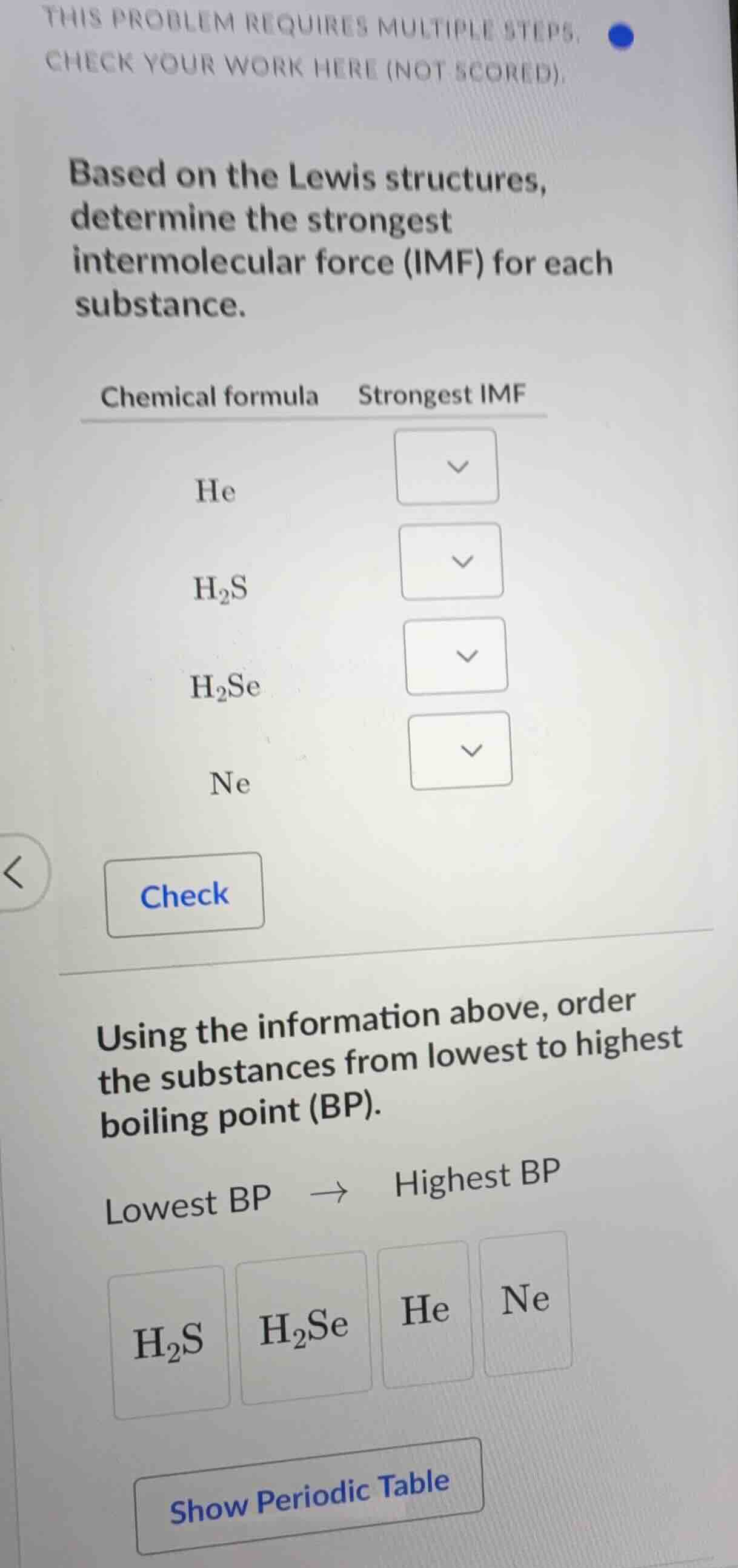
this problem requires multiple steps, check your work here (not scored). based on the lewis structures, determine the strongest intermolecular force (imf) for each substance. chemical formula strongest imf he h₂s h₂se ne using the information above, order the substances from lowest to highest boiling point (bp). lowest bp → highest bp h₂s h₂se he ne show periodic table
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
He: London dispersion forces
$\text{H}_2\text{S}$: Dipole-dipole interactions
$\text{H}_2\text{Se}$: Dipole-dipole interactions
Ne: London dispersion forces
He $\>$ Ne $\>$ $\text{H}_2\text{S}$ $\>$ $\text{H}_2\text{Se}$