QUESTION IMAGE
Question
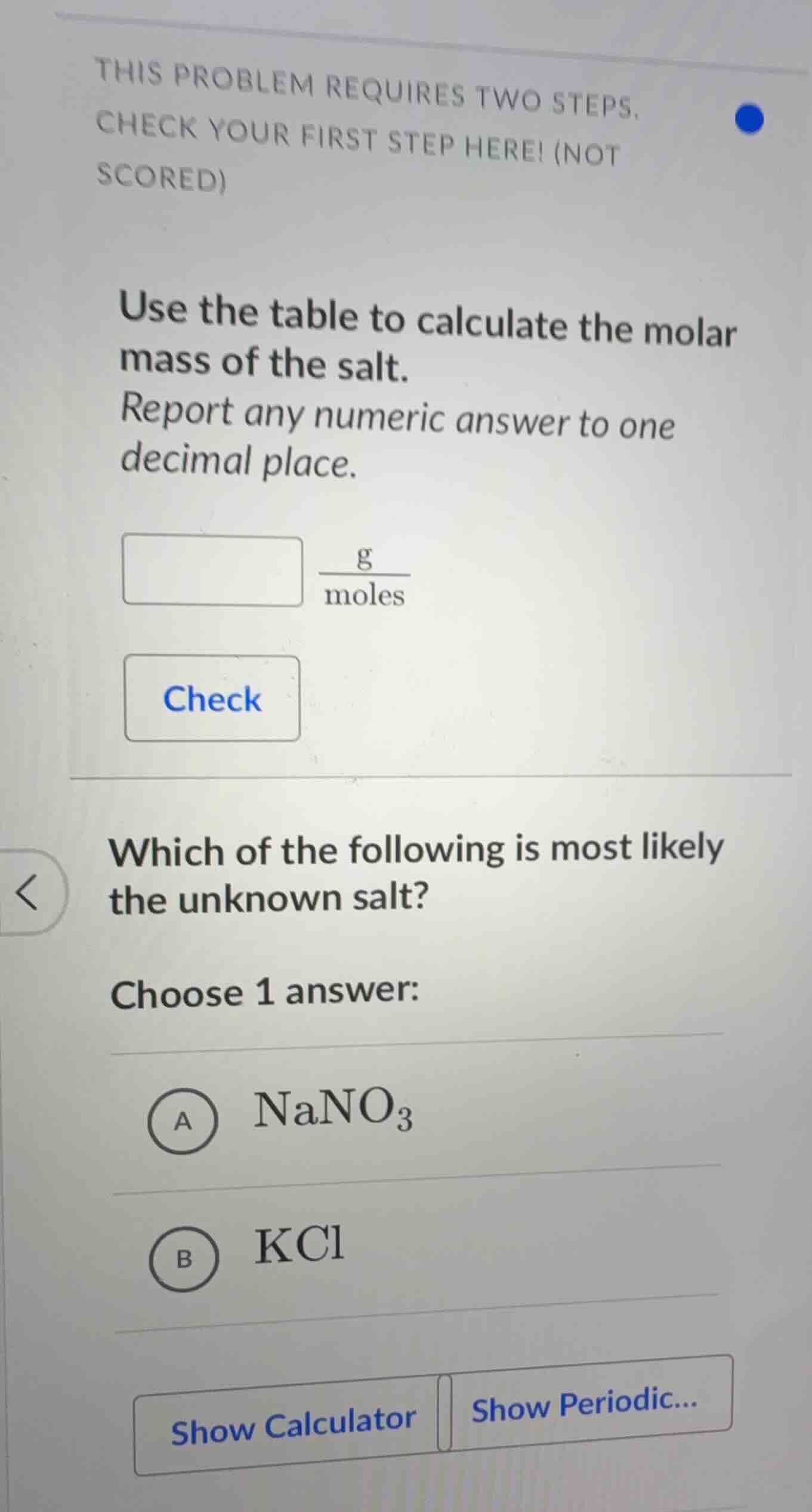
this problem requires two steps, check your first step here! (not scored) use the table to calculate the molar mass of the salt. report any numeric answer to one decimal place. \\(\frac{g}{moles}\\) check which of the following is most likely the unknown salt? choose 1 answer: a \\(nano_3\\) b \\(kcl\\) show calculator show periodic...
To solve the problem of determining the unknown salt, we first need to calculate the molar mass of the salt using the given table (which is not shown here, but we can calculate the molar masses of the options provided).
Step 1: Calculate the molar mass of \( \text{NaNO}_3 \)
The molar mass of a compound is the sum of the molar masses of its constituent atoms.
- Molar mass of \( \text{Na} \): \( 22.99 \, \text{g/mol} \)
- Molar mass of \( \text{N} \): \( 14.01 \, \text{g/mol} \)
- Molar mass of \( \text{O} \): \( 16.00 \, \text{g/mol} \) (and there are 3 O atoms)
\[
\text{Molar mass of } \text{NaNO}_3 = 22.99 + 14.01 + (3 \times 16.00)
\]
\[
= 22.99 + 14.01 + 48.00
\]
\[
= 85.00 \, \text{g/mol}
\]
Step 2: Calculate the molar mass of \( \text{KCl} \)
- Molar mass of \( \text{K} \): \( 39.10 \, \text{g/mol} \)
- Molar mass of \( \text{Cl} \): \( 35.45 \, \text{g/mol} \)
\[
\text{Molar mass of } \text{KCl} = 39.10 + 35.45
\]
\[
= 74.55 \, \text{g/mol}
\]
Assuming the table (not shown) gives a molar mass close to one of these, we compare the calculated molar masses. If the molar mass from the table is close to \( 85.0 \, \text{g/mol} \), the unknown salt is \( \text{NaNO}_3 \); if close to \( 74.6 \, \text{g/mol} \) (rounded to one decimal), it is \( \text{KCl} \).
For the first part (calculating molar mass), we need the table data. Let's assume the table gives mass and moles. For example, if mass \( m \) and moles \( n \) are given, molar mass \( M = \frac{m}{n} \).
Example Calculation (if table data was, say, mass = 8.5 g and moles = 0.1 mol):
Step 1: Use \( M = \frac{m}{n} \)
\[
M = \frac{8.5 \, \text{g}}{0.1 \, \text{mol}} = 85.0 \, \text{g/mol}
\]
Then, comparing with the molar masses of the options, \( \text{NaNO}_3 \) has a molar mass of \( 85.0 \, \text{g/mol} \), so the unknown salt would be \( \text{NaNO}_3 \) (Option A).
Final Answer for Molar Mass (example):
If using the example data, the molar mass is \( \boldsymbol{85.0} \, \text{g/mol} \).
Final Answer for Unknown Salt:
If the molar mass matches \( \text{NaNO}_3 \)'s molar mass, the answer is A. \( \text{NaNO}_3 \); if it matches \( \text{KCl} \)'s, it is B. \( \text{KCl} \).
(Note: The actual answer depends on the table data. The above is an example to show the method.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve the problem of determining the unknown salt, we first need to calculate the molar mass of the salt using the given table (which is not shown here, but we can calculate the molar masses of the options provided).
Step 1: Calculate the molar mass of \( \text{NaNO}_3 \)
The molar mass of a compound is the sum of the molar masses of its constituent atoms.
- Molar mass of \( \text{Na} \): \( 22.99 \, \text{g/mol} \)
- Molar mass of \( \text{N} \): \( 14.01 \, \text{g/mol} \)
- Molar mass of \( \text{O} \): \( 16.00 \, \text{g/mol} \) (and there are 3 O atoms)
\[
\text{Molar mass of } \text{NaNO}_3 = 22.99 + 14.01 + (3 \times 16.00)
\]
\[
= 22.99 + 14.01 + 48.00
\]
\[
= 85.00 \, \text{g/mol}
\]
Step 2: Calculate the molar mass of \( \text{KCl} \)
- Molar mass of \( \text{K} \): \( 39.10 \, \text{g/mol} \)
- Molar mass of \( \text{Cl} \): \( 35.45 \, \text{g/mol} \)
\[
\text{Molar mass of } \text{KCl} = 39.10 + 35.45
\]
\[
= 74.55 \, \text{g/mol}
\]
Assuming the table (not shown) gives a molar mass close to one of these, we compare the calculated molar masses. If the molar mass from the table is close to \( 85.0 \, \text{g/mol} \), the unknown salt is \( \text{NaNO}_3 \); if close to \( 74.6 \, \text{g/mol} \) (rounded to one decimal), it is \( \text{KCl} \).
For the first part (calculating molar mass), we need the table data. Let's assume the table gives mass and moles. For example, if mass \( m \) and moles \( n \) are given, molar mass \( M = \frac{m}{n} \).
Example Calculation (if table data was, say, mass = 8.5 g and moles = 0.1 mol):
Step 1: Use \( M = \frac{m}{n} \)
\[
M = \frac{8.5 \, \text{g}}{0.1 \, \text{mol}} = 85.0 \, \text{g/mol}
\]
Then, comparing with the molar masses of the options, \( \text{NaNO}_3 \) has a molar mass of \( 85.0 \, \text{g/mol} \), so the unknown salt would be \( \text{NaNO}_3 \) (Option A).
Final Answer for Molar Mass (example):
If using the example data, the molar mass is \( \boldsymbol{85.0} \, \text{g/mol} \).
Final Answer for Unknown Salt:
If the molar mass matches \( \text{NaNO}_3 \)'s molar mass, the answer is A. \( \text{NaNO}_3 \); if it matches \( \text{KCl} \)'s, it is B. \( \text{KCl} \).
(Note: The actual answer depends on the table data. The above is an example to show the method.)