QUESTION IMAGE
Question
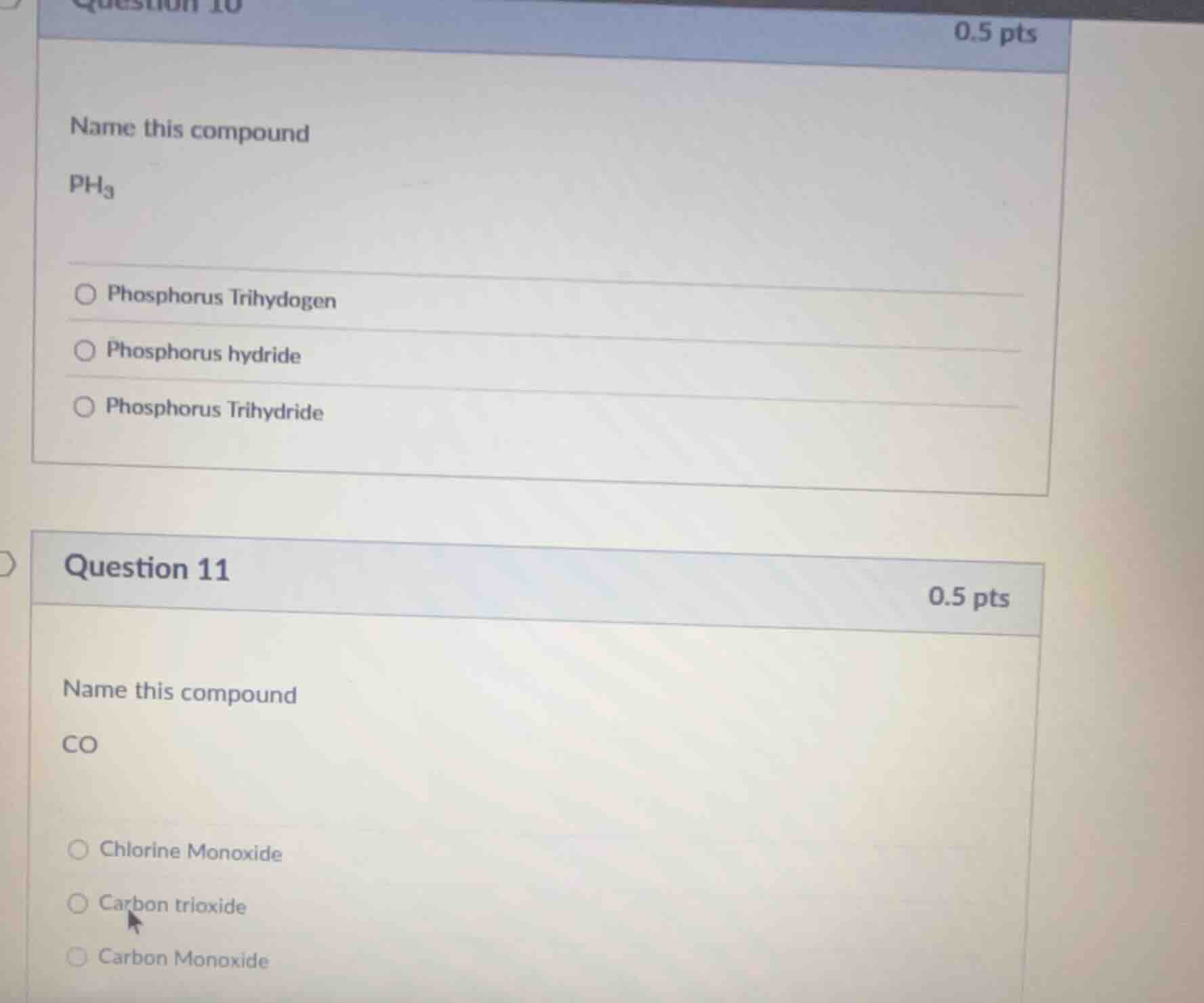
question 10
0.5 pts
name this compound
ph₃
○ phosphorus trihydogen
○ phosphorus hydride
○ phosphorus trihydride
question 11
0.5 pts
name this compound
co
○ chlorine monoxide
○ carbon trioxide
○ carbon monoxide
Question 10
To name the compound \( \text{PH}_3 \), we follow the rules for naming covalent compounds. The first element is phosphorus (P), and the second element is hydrogen (H). For covalent compounds, we use prefixes to indicate the number of atoms. There is 1 phosphorus atom (no prefix needed for the first element) and 3 hydrogen atoms, so we use the prefix "tri - " for hydrogen. The name of the compound is formed by naming the first element, then the second element with its ending changed to " - ide" and the appropriate prefix. So \( \text{PH}_3 \) is named Phosphorus Trihydride. The option "Phosphorus Trihydogen" has a misspelling (should be "hydride" not "hydogen"), and "Phosphorus hydride" does not use the prefix for the number of hydrogen atoms.
To name the compound \( \text{CO} \), we use the rules for covalent compounds. The first element is carbon (C), and the second element is oxygen (O). For covalent compounds, we use prefixes. There is 1 carbon atom (no prefix needed for the first element) and 1 oxygen atom. The prefix for 1 is "mono - " (used for the second element). The name of the compound is formed by naming the first element, then the second element with its ending changed to " - ide" and the appropriate prefix. So \( \text{CO} \) is named Carbon Monoxide. "Chlorine Monoxide" is incorrect because the first element is carbon, not chlorine. "Carbon trioxide" is incorrect because there is 1 oxygen atom, not 3.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Phosphorus Trihydride