QUESTION IMAGE
Question
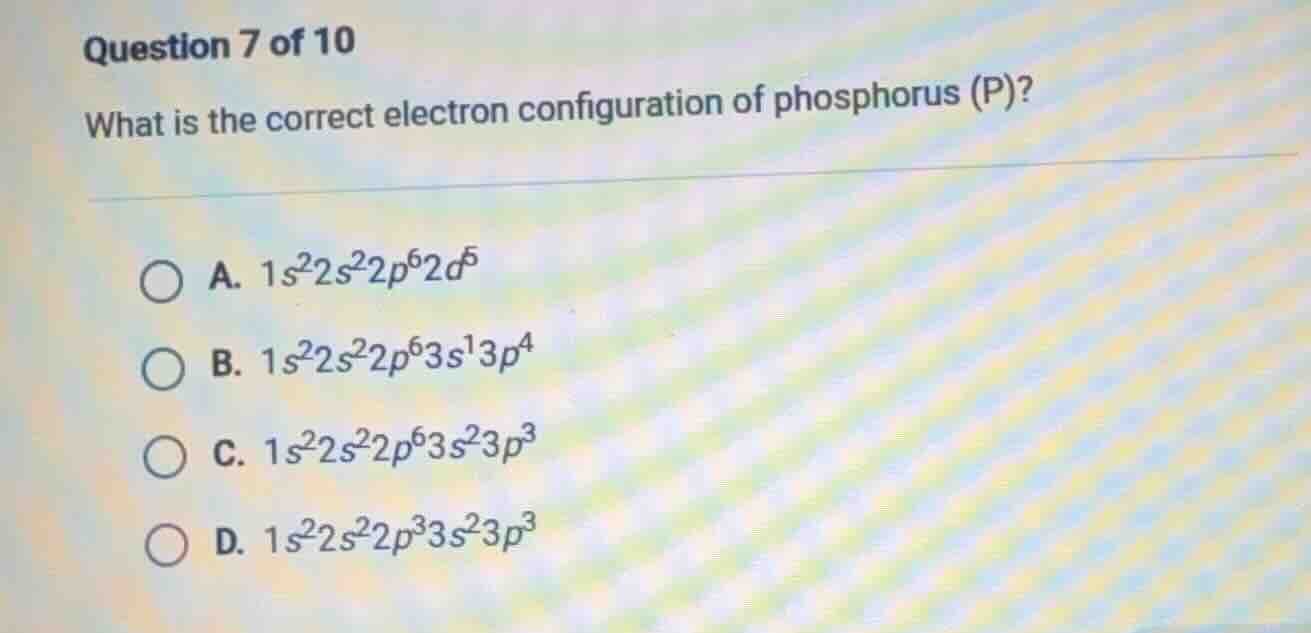
question 7 of 10
what is the correct electron configuration of phosphorus (p)?
a. $1s^2 2s^2 2p^6 2d^5$
b. $1s^2 2s^2 2p^6 3s^1 3p^4$
c. $1s^2 2s^2 2p^6 3s^2 3p^3$
d. $1s^2 2s^2 2p^3 3s^2 3p^3$
Brief Explanations
- First, recall the atomic number of phosphorus (P) is 15, so it has 15 electrons.
- Analyze each option:
- Option A: The \(2d\) subshell does not exist (electrons fill \(3s\) after \(2p\)), and the total electrons here are \(2 + 2+6 + 5=15\) but the subshell order is wrong.
- Option B: The sum of electrons is \(2 + 2+6 + 1+4 = 15\), but the filling of \(3s\) should be \(2\) electrons (not \(1\)) before filling \(3p\).
- Option C: Calculate total electrons: \(2+2 + 6+2 + 3=15\). The electron filling order is \(1s^22s^22p^63s^23p^3\) which follows the Aufbau principle (fill lower energy levels first, \(3s\) fills with 2 electrons before \(3p\) starts filling).
- Option D: The \(2p\) subshell should have 6 electrons (not 3) to be filled before moving to \(3s\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \(1s^22s^22p^63s^23p^3\)