QUESTION IMAGE
Question
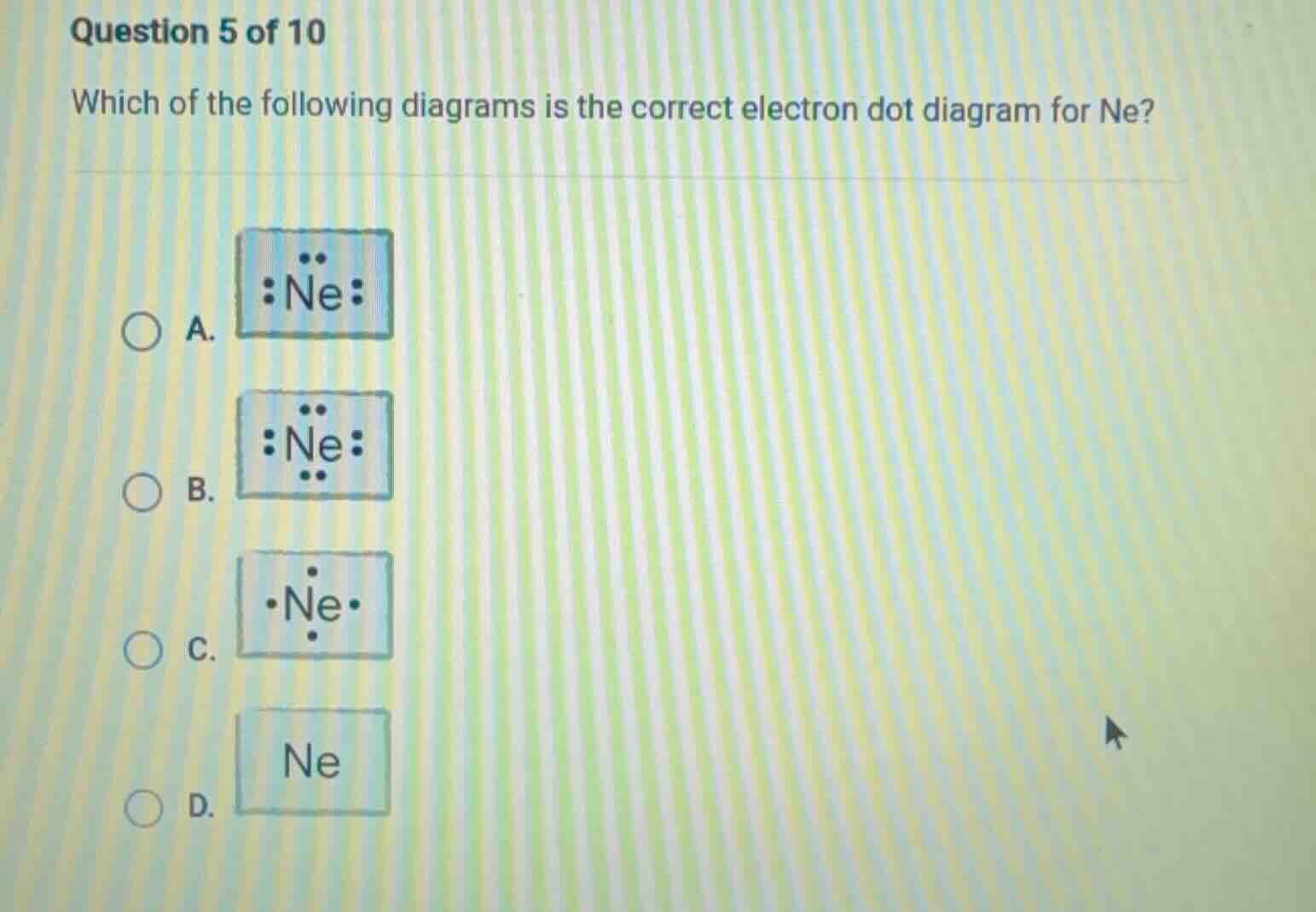
question 5 of 10
which of the following diagrams is the correct electron dot diagram for ne?
a.
:ne:
b.
:ne:
c.
·ne·
d.
ne
Neon (Ne) is a noble gas with an atomic number of 10, so it has 8 valence electrons (a stable octet). Electron dot diagrams (Lewis dot structures) represent valence electrons. For Ne, we place dots around the symbol to show 8 valence electrons. Option B has 8 dots (2 on top, 2 on right, 2 on bottom, 2 on left) around Ne, which matches the 8 valence electrons. Option A has 6 dots, C has 4, and D has none, so B is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\boxed{:\stackrel{..}{Ne}:}$ (or as per the diagram in option B: the diagram with 8 dots around Ne, 2 on each side)