QUESTION IMAGE
Question
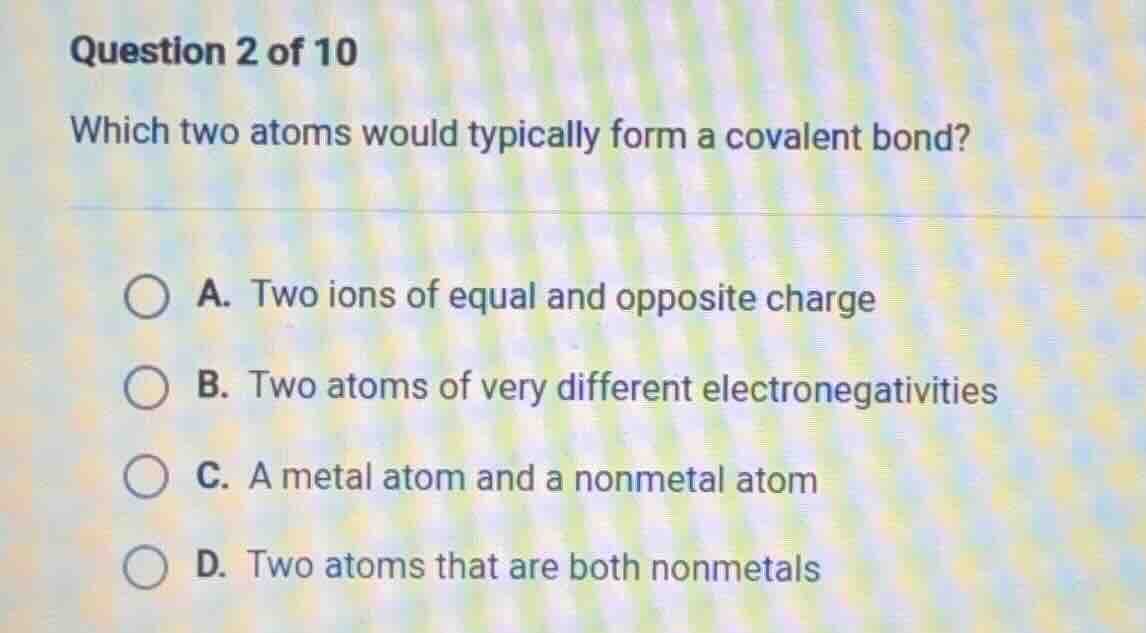
question 2 of 10
which two atoms would typically form a covalent bond?
a. two ions of equal and opposite charge
b. two atoms of very different electronegativities
c. a metal atom and a nonmetal atom
d. two atoms that are both nonmetals
Brief Explanations
To determine which two atoms form a covalent bond, we analyze each option:
- Option A: Ions with equal and opposite charge form ionic bonds (electrostatic attraction), not covalent.
- Option B: Atoms with very different electronegativities transfer electrons (ionic bond), not share (covalent).
- Option C: Metal - nonmetal combinations typically form ionic bonds (metal loses, nonmetal gains electrons).
- Option D: Covalent bonds involve sharing electrons, common between nonmetals (similar electronegativities, so they share rather than transfer electrons).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. Two atoms that are both nonmetals