QUESTION IMAGE
Question
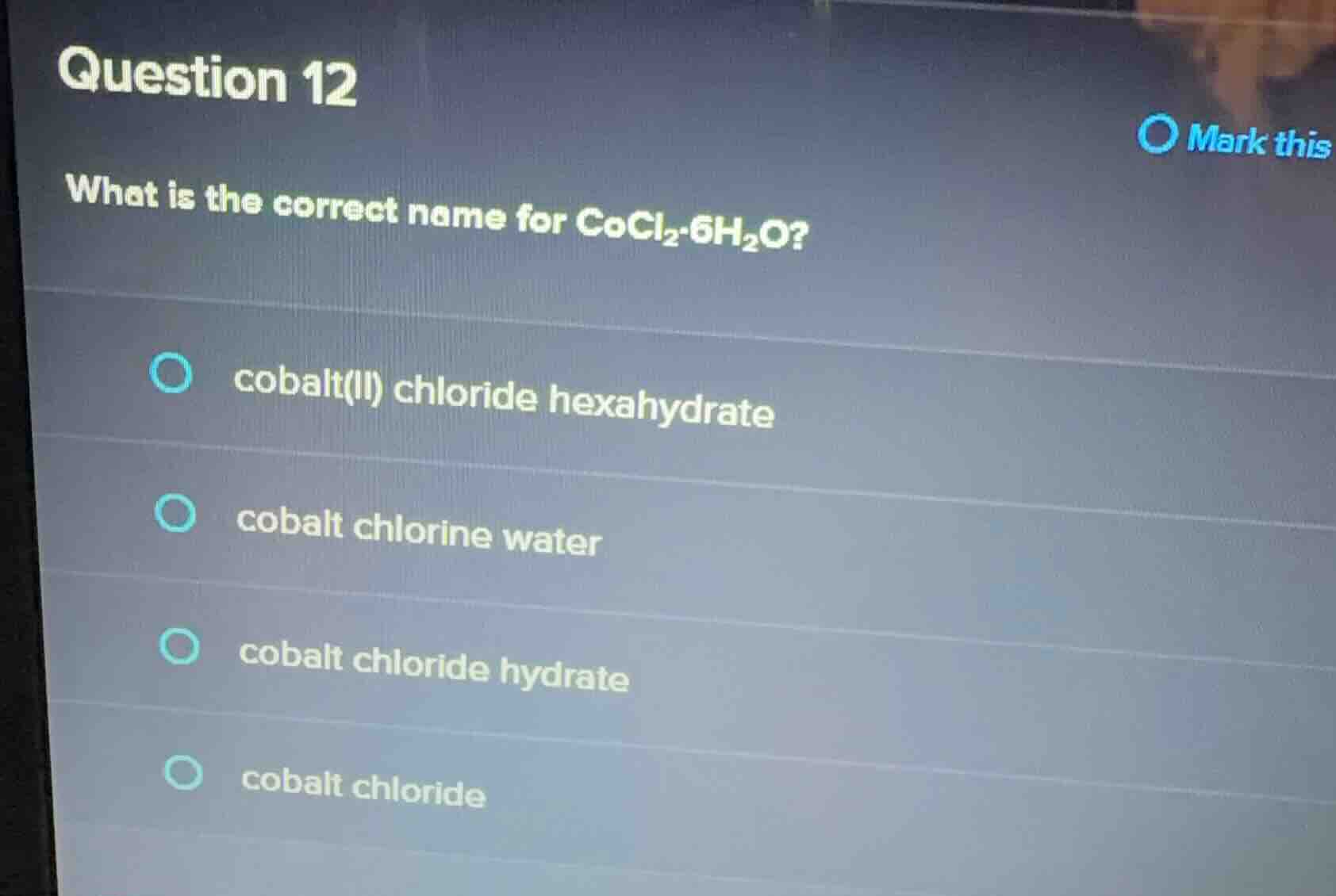
question 12
what is the correct name for cocl₂·6h₂o?
○ cobalt(ii) chloride hexahydrate
○ cobalt chlorine water
○ cobalt chloride hydrate
○ cobalt chloride
Brief Explanations
- First, identify the ionic compound: $\text{CoCl}_2$. Chloride has a -1 charge, so two chlorides give a total -2 charge. Cobalt must have a +2 charge to balance this, so we name it cobalt(II) chloride.
- Next, identify the water of hydration: $\text{6H}_2\text{O}$. The prefix for 6 is "hexa-", and we use the suffix "-hydrate" for water molecules in a hydrate.
- Combine the two parts: the ionic compound name followed by the hydrate name.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
cobalt(II) chloride hexahydrate