QUESTION IMAGE
Question
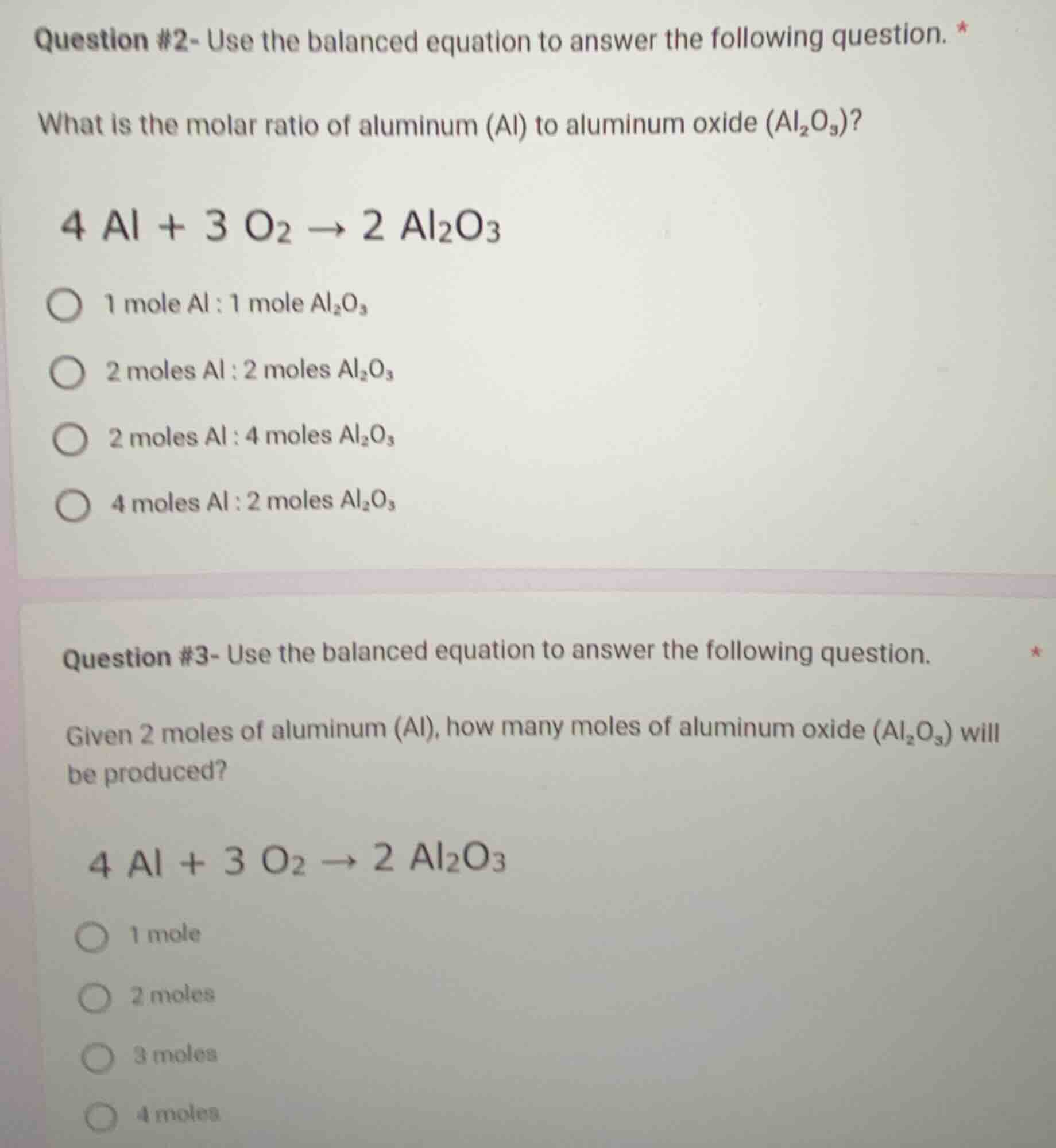
question #2- use the balanced equation to answer the following question. *
what is the molar ratio of aluminum (al) to aluminum oxide (al₂o₃)?
4 al + 3 o₂ → 2 al₂o₃
1 mole al : 1 mole al₂o₃
2 moles al : 2 moles al₂o₃
2 moles al : 4 moles al₂o₃
4 moles al : 2 moles al₂o₃
question #3- use the balanced equation to answer the following question. *
given 2 moles of aluminum (al), how many moles of aluminum oxide (al₂o₃) will be produced?
4 al + 3 o₂ → 2 al₂o₃
1 mole
2 moles
3 moles
4 moles
Step1: Identify Al and $\text{Al}_2\text{O}_3$ coefficients
From balanced equation: $4\text{Al} + 3\text{O}_2
ightarrow 2\text{Al}_2\text{O}_3$, coefficients are 4 (Al) and 2 ($\text{Al}_2\text{O}_3$).
Step2: Set molar ratio for Q2
Molar ratio = $\text{Moles of Al}:\text{Moles of }\text{Al}_2\text{O}_3 = 4:2$
Step3: Use ratio for Q3 mole calculation
Mole ratio: $\frac{\text{Moles of }\text{Al}_2\text{O}_3}{\text{Moles of Al}} = \frac{2}{4} = \frac{1}{2}$
Moles of $\text{Al}_2\text{O}_3 = 2 \times \frac{1}{2} = 1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question #2: 4 moles Al : 2 moles $\text{Al}_2\text{O}_3$
Question #3: 1 mole