QUESTION IMAGE
Question
question 23 of 37
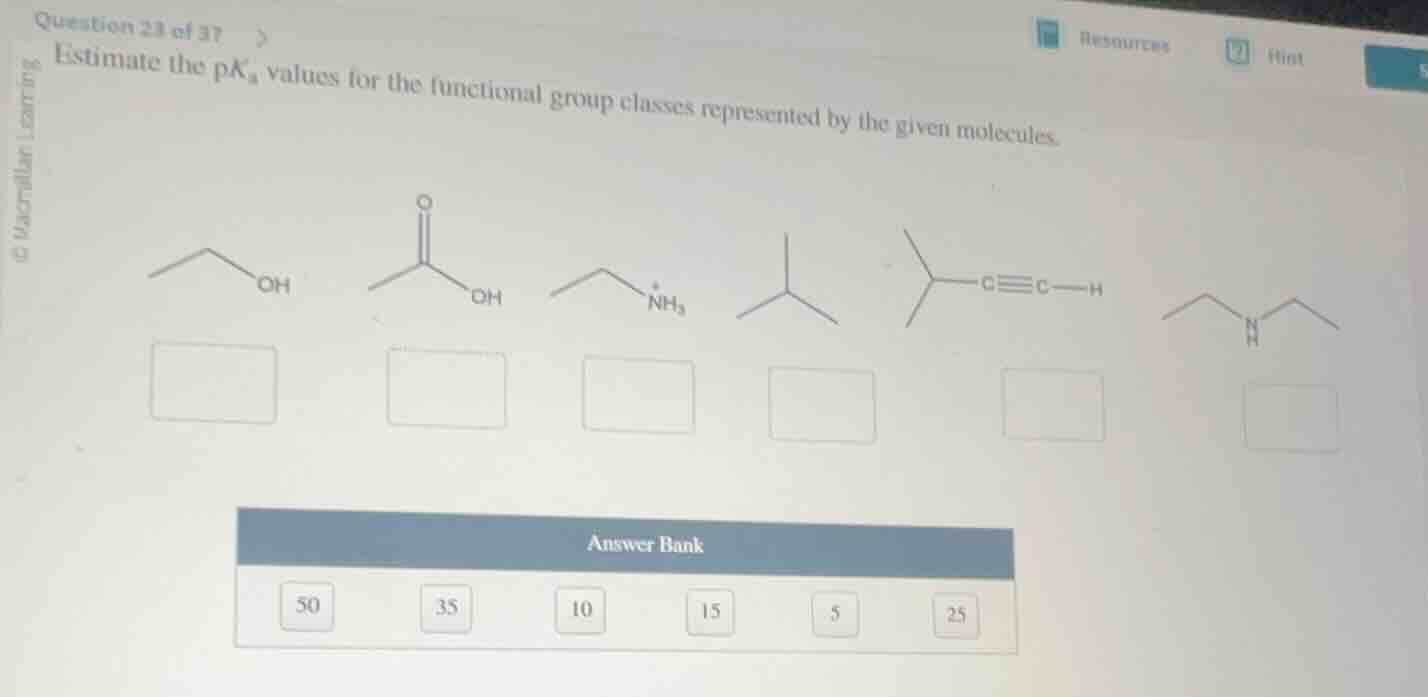
estimate the pkₐ values for the functional group classes represented by the given molecules.
(chemical structures of molecules and answer bank with numbers 50, 35, 10, 15, 5, 25)
Brief Explanations
- Ethanol (alcohol): Alcohols have typical pKa ~15.
- Acetic acid (carboxylic acid): Carboxylic acids have pKa ~5.
- Ethanammonium (protonated amine): Protonated aliphatic amines have pKa ~10.
- Isobutane (alkane): Alkanes are very weak acids, pKa ~50.
- 3-methyl-1-butyne (terminal alkyne): Terminal alkynes have pKa ~25.
- Diethylamine (amine, neutral): Neutral aliphatic amines have pKa ~35 (pKa of their conjugate acid is ~10, so pKa of the free amine is much higher).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Ethanol: 15
- Acetic acid: 5
- Ethanammonium: 10
- Isobutane: 50
- 3-methyl-1-butyne: 25
- Diethylamine: 35