QUESTION IMAGE
Question
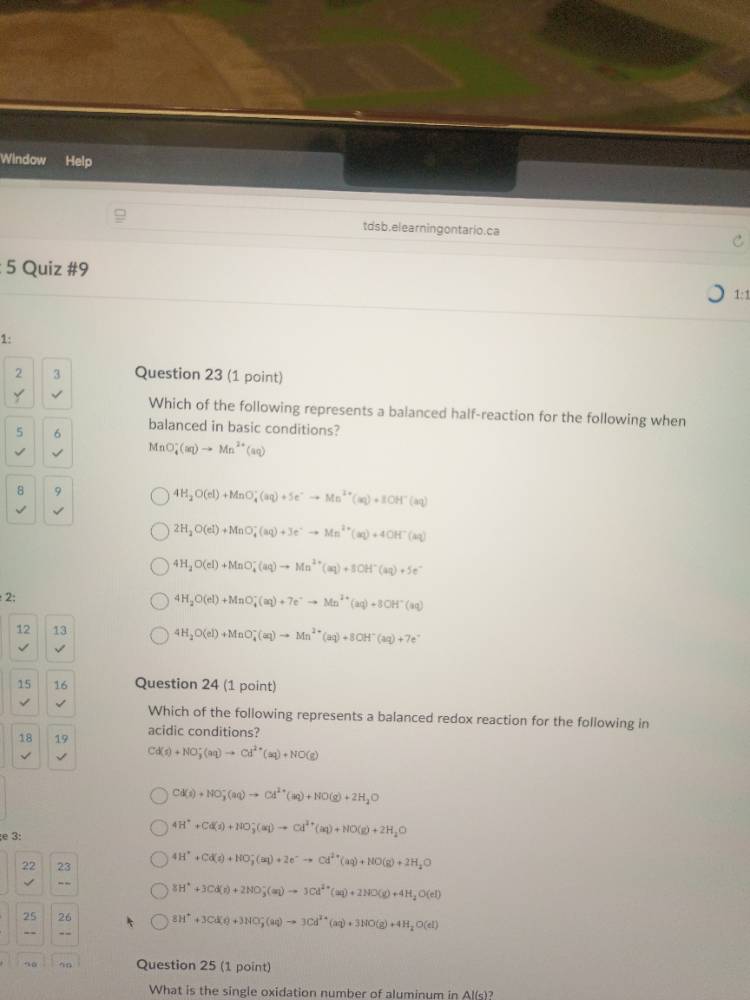
question 23 (1 point)
which of the following represents a balanced half - reaction for the following when balanced in basic conditions?
mno₄⁻(aq) → mn²⁺(aq)
① 4h₂o(l) + mno₄⁻(aq) + 5e⁻ → mn²⁺(aq) + 8oh⁻(aq)
② 2h₂o(l) + mno₄⁻(aq) + 3e⁻ → mn²⁺(aq) + 4oh⁻(aq)
③ 4h₂o(l) + mno₄⁻(aq) → mn²⁺(aq) + 8oh⁻(aq) + 5e⁻
④ 4h₂o(l) + mno₄⁻(aq) + 7e⁻ → mn²⁺(aq) + 8oh⁻(aq)
⑤ 4h₂o(l) + mno₄⁻(aq) → mn²⁺(aq) + 8oh⁻(aq) + 7e⁻
question 24 (1 point)
which of the following represents a balanced redox reaction for the following in acidic conditions?
cd(s) + no₃⁻(aq) → cd²⁺(aq) + no(g)
① cd(s) + no₃⁻(aq) → cd²⁺(aq) + no(g) + 2h₂o
② 4h⁺ + cd(s) + no₃⁻(aq) → cd²⁺(aq) + no(g) + 2h₂o
③ 4h⁺ + cd(s) + no₃⁻(aq) + 2e⁻ → cd²⁺(aq) + no(g) + 2h₂o
④ 8h⁺ + 3cd(s) + 2no₃⁻(aq) → 3cd²⁺(aq) + 2no(g) + 4h₂o(l)
⑤ 8h⁺ + 3cd(s) + 3no₃⁻(aq) → 3cd²⁺(aq) + 3no(g) + 4h₂o(l)
question 25 (1 point)
what is the single oxidation number of aluminum in al(s)?
Question 23
Step1: Determine oxidation state change
Mn in \(MnO_4^-(aq)\) has an oxidation state of +7 (since O is -2, \(x + 4(-2)= -1\) → \(x = +7\)), and in \(Mn^{2+}(aq)\) it is +2. So the change in oxidation state is \(+7 \to +2\), which is a gain of 5 electrons (reduction, so electrons are on the left in basic conditions).
Step2: Balance O by adding \(H_2O\)
\(MnO_4^-(aq)\) has 4 O atoms, so add \(4H_2O(l)\) to the right? Wait, no, in basic conditions, to balance O, we first balance O by adding \(H_2O\) to the side with less O. Wait, \(MnO_4^-\) has 4 O, so to balance O, we add \(4H_2O(l)\) to the left? No, wait, the product is \(Mn^{2+}\), which has no O. So we need to balance O by adding \(H_2O\) to the side without O (right) and \(OH^-\) to the left? Wait, no, the correct method for basic conditions:
- Balance atoms other than O and H: Mn is balanced (1 Mn on each side).
- Balance O by adding \(H_2O\): \(MnO_4^-\) has 4 O, so add \(4H_2O(l)\) to the right (product side) to balance O: \(MnO_4^-(aq) \to Mn^{2+}(aq) + 4H_2O(l)\)
- Balance H by adding \(H^+\) to the side with less H, then add \(OH^-\) to both sides to neutralize \(H^+\) (since it's basic). Wait, the right side has \(4H_2O\), which has 8 H. So add \(8H^+(aq)\) to the left: \(8H^+(aq) + MnO_4^-(aq) \to Mn^{2+}(aq) + 4H_2O(l)\)
- Now, add \(8OH^-(aq)\) to both sides to neutralize \(H^+\): \(8OH^-(aq) + 8H^+(aq) + MnO_4^-(aq) \to Mn^{2+}(aq) + 4H_2O(l) + 8OH^-(aq)\)
- Simplify \(H^+\) and \(OH^-\) to \(H_2O\): \(8H_2O(l) + MnO_4^-(aq) \to Mn^{2+}(aq) + 4H_2O(l) + 8OH^-(aq)\)
- Subtract \(4H_2O(l)\) from both sides: \(4H_2O(l) + MnO_4^-(aq) \to Mn^{2+}(aq) + 8OH^-(aq)\)
- Now balance charge: Left side charge: \(MnO_4^-\) is -1, \(4H_2O\) is neutral, so total -1. Right side: \(Mn^{2+}\) is +2, \(8OH^-\) is -8, so total \(2 - 8 = -6\). Wait, no, we missed the electrons. Wait, the oxidation state change is 5 electrons gained (since Mn goes from +7 to +2, gain of 5 e⁻). So add 5 e⁻ to the left: \(4H_2O(l) + MnO_4^-(aq) + 5e^- \to Mn^{2+}(aq) + 8OH^-(aq)\)
Wait, let's check the options. The first option is \(4H_2O(l) + MnO_4^-(aq) + 5e^- \to Mn^{2+}(aq) + 8OH^-(aq)\), which matches our calculation. Let's verify:
- Mn: 1 on left, 1 on right.
- O: 4 (from \(MnO_4^-\)) + 4 (from \(4H_2O\))? Wait no, \(4H_2O\) has 4 O, \(MnO_4^-\) has 4 O? Wait no, \(4H_2O\) has 4 O, \(MnO_4^-\) has 4 O, so left side O: 4 (from \(MnO_4^-\)) + 4 (from \(4H_2O\))? No, wait the first option is \(4H_2O(l) + MnO_4^-(aq) + 5e^- \to Mn^{2+}(aq) + 8OH^-(aq)\). Let's count O:
Left: \(4H_2O\) has 4 O, \(MnO_4^-\) has 4 O? No, \(4H_2O\) is 4 O, \(MnO_4^-\) is 4 O? Wait no, \(4H_2O\) is 4 O, \(MnO_4^-\) is 4 O, so total 8 O? Right side: \(8OH^-\) has 8 O. So O is balanced.
H: Left: \(4H_2O\) has 8 H. Right: \(8OH^-\) has 8 H (since each \(OH^-\) has 1 H, 8*1=8). So H is balanced.
Charge: Left: \(MnO_4^-\) is -1, \(5e^-\) is -5, \(4H_2O\) is neutral, so total -6. Right: \(Mn^{2+}\) is +2, \(8OH^-\) is -8, so total -6. Charge is balanced.
Electrons: Gain of 5 e⁻ (reduction), which matches the oxidation state change (Mn from +7 to +2, gain 5 e⁻). So the first option is correct.
We need to balance the redox reaction \(Cd(s) + NO_3^-(aq) \to Cd^{2+}(aq) + NO(g)\) in acidic conditions.
Step 1: Identify oxidation and reduction half-reactions
- Oxidation: \(Cd(s) \to Cd^{2+}(aq)\) (Cd goes from 0 to +2, loss of 2 electrons)
- Reduction: \(NO_3^-(aq) \to NO(g)\) (N goes from +5 to +2, gain of 3 electrons)
Step 2: Balance each half-reaction in acidic conditions
- Oxidation half-reaction (Cd):
- Balance Cd: \(Cd(s) \to Cd^{2+}(aq)\)
- Balance charge: Add 2 electrons (since it's oxidation, electrons on right): \(Cd(s) \to Cd^{2+}(aq) + 2e^-\)
- Reduction half-reaction (NO₃⁻):
- Balance N: \(NO_3^-(aq) \to NO(g)\)
- Balance O: Add \(2H_2O(l)\) to the right (since NO has 1 O, NO₃⁻ has 3 O; 3 - 1 = 2 O, so add 2 H₂O to right): \(NO_3^-(aq) \to NO(g) + 2H_2O(l)\)
- Balance H: Add \(4H^+(aq)\) to the left (since right has 4 H from 2 H₂O): \(4H^+(aq) + NO_3^-(aq) \to NO(g) + 2H_2O(l)\)
- Balance charge: Left charge: \(4H^+\) (+4) + \(NO_3^-\) (-1) = +3. Right charge: NO (0) + 2 H₂O (0) = 0. So gain of 3 electrons (add to left): \(4H^+(aq) + NO_3^-(aq) + 3e^- \to NO(g) + 2H_2O(l)\)
Step 3: Equalize electrons gained and lost
Oxidation: \(Cd(s) \to Cd^{2+}(aq) + 2e^-\) (multiply by 3: \(3Cd(s) \to 3Cd^{2+}(aq) + 6e^-\))
Reduction: \(4H^+(aq) + NO_3^-(aq) + 3e^- \to NO(g) + 2H_2O(l)\) (multiply by 2: \(8H^+(aq) + 2NO_3^-(aq) + 6e^- \to 2NO(g) + 4H_2O(l)\))
Step 4: Add the two half-reactions
\(3Cd(s) + 8H^+(aq) + 2NO_3^-(aq) + 6e^- \to 3Cd^{2+}(aq) + 6e^- + 2NO(g) + 4H_2O(l)\)
Cancel electrons: \(3Cd(s) + 8H^+(aq) + 2NO_3^-(aq) \to 3Cd^{2+}(aq) + 2NO(g) + 4H_2O(l)\)
Wait, let's check the options. The fifth option (last one) is \(8H^+ + 3Cd(s) + 3NO_3^-(aq) \to 3Cd^{2+}(aq) + 3NO(g) + 4H_2O(l)\)? No, wait the options:
Wait the options are:
- \(Cd(s) + NO_3^-(aq) \to Cd^{2+}(aq) + NO(g) + 2H_2O\) – electrons not balanced, charge not balanced.
- \(4H^+ + Cd(s) + NO_3^-(aq) \to Cd^{2+}(aq) + NO(g) + 2H_2O\) – electrons? Let's check charge: left: 4+ + 0 + (-1) = +3; right: +2 + 0 + 0 = +2. Not balanced.
- \(4H^+ + Cd(s) + NO_3^-(aq) + 2e^- \to Cd^{2+}(aq) + NO(g) + 2H_2O\) – electrons on left (reduction), but Cd is oxidation (electrons on right). Mixed, wrong.
- \(8H^+ + 3Cd(s) + 2NO_3^-(aq) \to 3Cd^{2+}(aq) + 2NO(g) + 4H_2O(l)\) – this matches our balanced equation (3 Cd, 8 H⁺, 2 NO₃⁻, 3 Cd²⁺, 2 NO, 4 H₂O). Wait the fourth option (before last) is \(8H^+ + 3Cd(s) + 2NO_3^-(aq) \to 3Cd^{2+}(aq) + 2NO(g) + 4H_2O(l)\), which is our balanced equation. Wait the last option is \(8H^+ + 3Cd(s) + 3NO_3^-(aq) \to 3Cd^{2+}(aq) + 3NO(g) + 4H_2O(l)\) – no, that has 3 NO₃⁻, which would give 3 NO, but our calculation has 2 NO₃⁻ giving 2 NO. Wait maybe I made a mistake in multiplying.
Wait oxidation: 2 e⁻ lost per Cd, reduction: 3 e⁻ gained per NO₃⁻. So LCM of 2 and 3 is 6. So oxidation: 3 Cd (loses 6 e⁻), reduction: 2 NO₃⁻ (gains 6 e⁻). So the balanced equation should have 3 Cd, 2 NO₃⁻, 8 H⁺ (from 24 H⁺), 3 Cd²⁺, 2 NO, 4 H₂O (from 22 H₂O). So the fourth option (the one with \(8H^+ + 3Cd(s) + 2NO_3^-(aq) \to 3Cd^{2+}(aq) + 2NO(g) + 4H_2O(l)\)) is correct.
Wait let's check charge:
Left: 8H⁺ (+8) + 3Cd (0) + 2NO₃⁻ (-2) = +6.
Right: 3Cd²⁺ (+6) + 2NO (0) + 4H₂O (0) = +6. Charge balanced.
Atoms:
- Cd: 3 on left, 3 on right.
- N: 2 on left (2 NO₃⁻), 2 on right (2 NO).
- O: 23 (6 from 2 NO₃⁻) = 6; right: 21 (2 from 2 NO) + 4*1 (4 from 4 H₂O) = 6. Balanced.
- H: 8 on left (8 H⁺), 4*2 (8 from 4 H₂O) on right. Balanced.
So the correct option is the fourth one (the one with \(8H^+ + 3Cd(…
In \(Al(s)\) (solid aluminum), the oxidation number of an element in its elemental form is 0. So aluminum in \(Al(s)\) has an oxidation number of 0.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \(4H_2O(l) + MnO_4^-(aq) + 5e^- \to Mn^{2+}(aq) + 8OH^-(aq)\) (assuming the first option is labeled A, but in the image, the first option is the one with \(4H_2O\), \(MnO_4^-\), \(5e^-\), etc.)