QUESTION IMAGE
Question
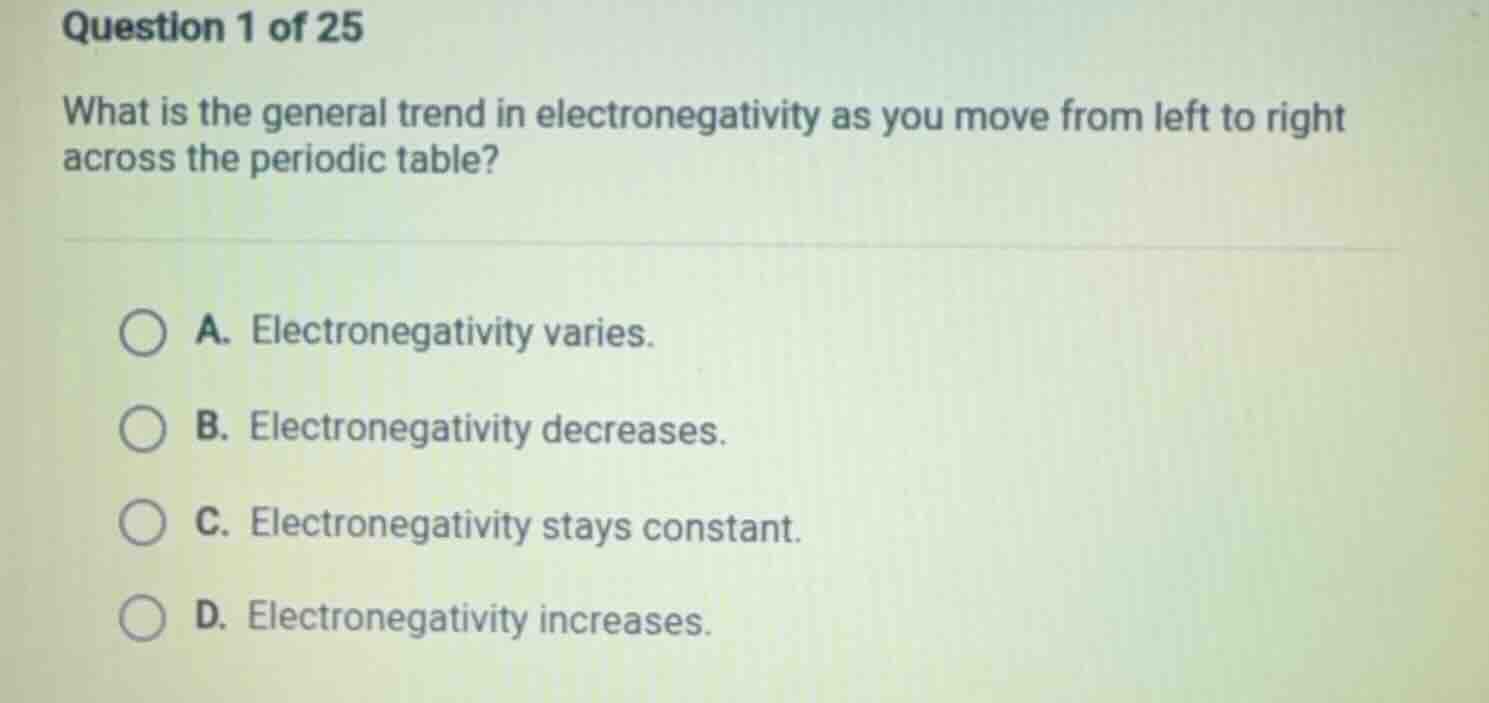
question 1 of 25
what is the general trend in electronegativity as you move from left to right across the periodic table?
a. electronegativity varies.
b. electronegativity decreases.
c. electronegativity stays constant.
d. electronegativity increases.
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. As we move from left to right across a period (row) in the periodic table, the number of protons in the nucleus increases (effective nuclear charge increases) while the shielding effect from inner electrons remains relatively constant. This increased positive charge in the nucleus pulls the valence electrons more strongly, leading to an increase in electronegativity. So the general trend as we move left to right across the periodic table is that electronegativity increases.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. Electronegativity increases.