QUESTION IMAGE
Question
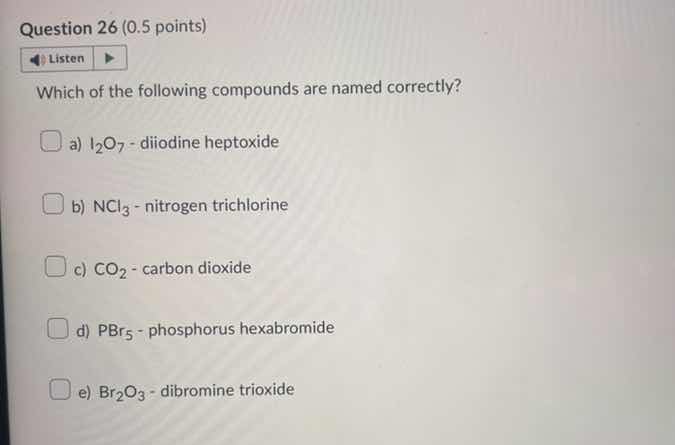
question 26 (0.5 points) listen which of the following compounds are named correctly? a) $\ce{i_{2}o_{7}}$ - diiodine heptoxide b) $\ce{ncl_{3}}$ - nitrogen trichlorine c) $\ce{co_{2}}$ - carbon dioxide d) $\ce{pbr_{5}}$ - phosphorus hexabromide e) $\ce{br_{2}o_{3}}$ - dibromine trioxide
Brief Explanations
- For binary covalent compounds, the first element uses its full name, the second uses a prefix + root + "-ide". Prefixes match the subscript: 1=mono (omitted for first element), 2=di, 3=tri, 4=tetra, 5=penta, 6=hexa, 7=hepta.
- a) $\text{I}_2\text{O}_7$: Correct prefixes (di- for 2 I, hepta- for 7 O) + -ide for oxygen: diiodine heptoxide.
- b) $\text{NCl}_3$: Should be nitrogen trichloride (ends with -ide, not "trichlorine").
- c) $\text{CO}_2$: Correct (carbon + di- + oxide root + -ide: carbon dioxide).
- d) $\text{PBr}_5$: Subscript 5 uses "penta-", not "hexa-": should be phosphorus pentabromide.
- e) $\text{Br}_2\text{O}_3$: Correct prefixes (di- for 2 Br, tri- for 3 O) + -ide: dibromine trioxide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) $\text{I}_2\text{O}_7$ - diiodine heptoxide
c) $\text{CO}_2$ - carbon dioxide
e) $\text{Br}_2\text{O}_3$ - dibromine trioxide