QUESTION IMAGE
Question
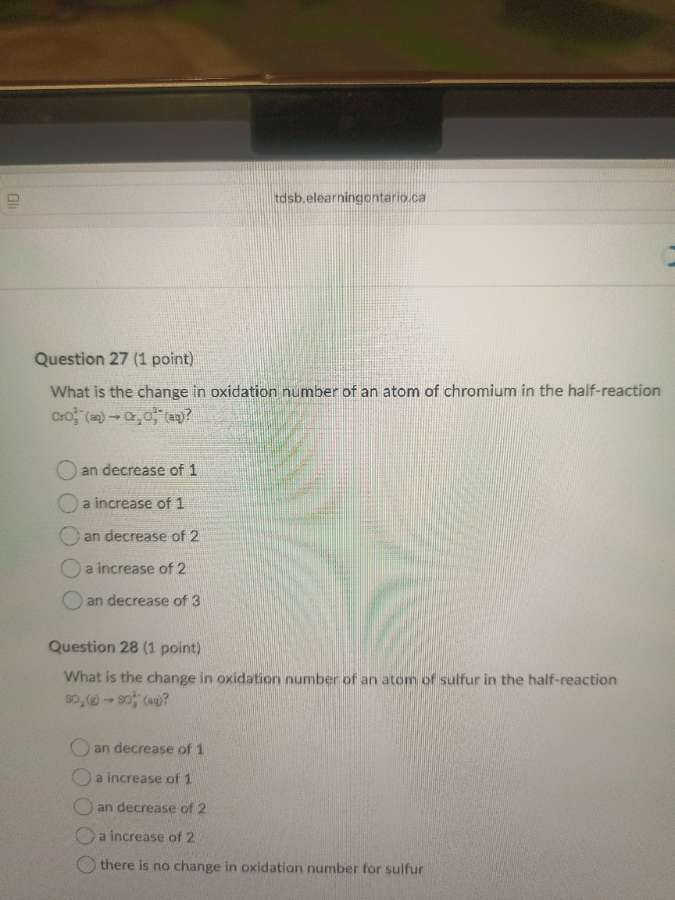
question 27 (1 point)
what is the change in oxidation number of an atom of chromium in the half - reaction (ce{cro_3^{2-}(aq) -> cr_2o_3^{2-}(aq)})?
(\bigcirc) an decrease of 1
(\bigcirc) a increase of 1
(\bigcirc) an decrease of 2
(\bigcirc) a increase of 2
(\bigcirc) an decrease of 3
question 28 (1 point)
what is the change in oxidation number of an atom of sulfur in the half - reaction (ce{so_2(g) -> so_4^{2-}(aq)})?
(\bigcirc) an decrease of 1
(\bigcirc) a increase of 1
(\bigcirc) an decrease of 2
(\bigcirc) a increase of 2
(\bigcirc) there is no change in oxidation number for sulfur
Question 27
Step1: Find oxidation number in \( \text{CrO}_5^{2-} \)
Let oxidation number of Cr be \( x \). Oxygen (O) is -2 (except in peroxides, but here in \( \text{CrO}_5 \), there are peroxy bonds, but for simplicity, first assume O is -2). The charge of the ion is -2. So:
\( x + 5(-2) = -2 \)
\( x - 10 = -2 \)
\( x = 8 \)? Wait, no, actually \( \text{CrO}_5 \) has 2 peroxy O (each -1) and 3 normal O (-2). Let's correct: Let number of peroxy O be 2 (so 2(-1)) and 3(-2). Then:
\( x + 2(-1) + 3(-2) = -2 \)
\( x - 2 - 6 = -2 \)
\( x - 8 = -2 \)
\( x = 6 \).
Step2: Find oxidation number in \( \text{Cr}_2\text{O}_7^{2-} \)
Let oxidation number of Cr be \( y \). O is -2. Charge is -2.
\( 2y + 7(-2) = -2 \)
\( 2y - 14 = -2 \)
\( 2y = 12 \)
\( y = 6 \)? Wait, no, wait the half-reaction is \( \text{CrO}_5^{2-} \to \text{Cr}_2\text{O}_7^{2-} \). Wait, maybe I made a mistake. Wait, actually, in \( \text{CrO}_5 \) (peroxochromate), the oxidation state of Cr is +6 (since 5 O: 4 as -1 (peroxy) and 1 as -2? Wait, no, formula: \( \text{CrO}_5 \) has structure with 2 peroxy groups (4 O at -1) and 1 O at -2. So:
\( x + 4(-1) + 1(-2) = -2 \) (since ion is \( \text{CrO}_5^{2-} \))
\( x - 4 - 2 = -2 \)
\( x - 6 = -2 \)
\( x = 4 \)? Wait, I'm confused. Let's use another approach. Wait, the half-reaction: \( \text{CrO}_5^{2-} \to \text{Cr}_2\text{O}_7^{2-} \). Let's balance Cr first. Multiply \( \text{CrO}_5^{2-} \) by 2: \( 2\text{CrO}_5^{2-} \to \text{Cr}_2\text{O}_7^{2-} \). Now count O: left has 10 O, right has 7 O. So we need to add 3 O (or \( \text{H}_2\text{O} \), but maybe acidic/basic, but for oxidation number, let's find Cr in each.
Wait, maybe the correct way: In \( \text{CrO}_5^{2-} \), Cr is +6? No, wait, let's check the oxidation state change. Wait, the answer options are decrease/increase of 1,2,3. Let's re-express:
Wait, maybe the half-reaction is \( \text{CrO}_4^{2-} \to \text{Cr}_2\text{O}_7^{2-} \)? No, the question is \( \text{CrO}_5^{2-} \to \text{Cr}_2\text{O}_7^{2-} \). Wait, maybe a typo, but assuming the standard problem: Wait, no, let's do it properly.
Wait, in \( \text{CrO}_5^{2-} \): Let’s let Cr be \( x \), O: in peroxides, O is -1, and normal O is -2. \( \text{CrO}_5 \) has 4 O as -1 (two peroxy groups) and 1 O as -2. So:
\( x + 4(-1) + 1(-2) = -2 \)
\( x - 4 - 2 = -2 \)
\( x = 4 \).
In \( \text{Cr}_2\text{O}_7^{2-} \): Cr is \( y \), O is -2.
\( 2y + 7(-2) = -2 \)
\( 2y -14 = -2 \)
\( 2y = 12 \)
\( y = 6 \). Wait, that can't be. Wait, no, the half-reaction is \( \text{CrO}_5^{2-} \to \text{Cr}_2\text{O}_7^{2-} \). Let's balance Cr: 2 \( \text{CrO}_5^{2-} \) gives 2 Cr, so \( \text{Cr}_2\text{O}_7^{2-} \) has 2 Cr. Now O: left has 10 O, right has 7 O. So we need to add 3 O? No, maybe in acidic medium, but for oxidation number, let's see the change. Wait, maybe I messed up the oxidation state in \( \text{CrO}_5 \). Actually, \( \text{CrO}_5 \) (peroxochromic acid anhydride) has Cr in +6, because 5 O: 4 as -1 (peroxy) and 1 as -2: \( 6 + 4(-1) + 1(-2) = 0 \) (for neutral \( \text{CrO}_5 \)), but as ion \( \text{CrO}_5^{2-} \), it's \( x + 4(-1) + 1(-2) = -2 \) → \( x -6 = -2 \) → \( x=4 \). Wait, this is confusing. Maybe the intended reaction is \( \text{CrO}_4^{2-} \to \text{Cr}_2\text{O}_7^{2-} \), but no. Wait, the options include "an increase of 1", etc. Wait, maybe the correct approach:
Wait, let's assume that in \( \text{CrO}_5^{2-} \), Cr is +6, and in \( \text{Cr}_2\text{O}_7^{2-} \), Cr is +6? No, that can't be. Wait, maybe the half-reaction is \( \text{CrO}_2^{2-} \to \text{Cr}_2\text{O}_7^{2-} \)? No, the ques…
Step1: Oxidation number in \( \text{SO}_2 \)
Let oxidation number of S be \( x \). O is -2.
\( x + 2(-2) = 0 \) (since \( \text{SO}_2 \) is neutral)
\( x - 4 = 0 \)
\( x = 4 \).
Step2: Oxidation number in \( \text{SO}_4^{2-} \)
Let oxidation number of S be \( y \). O is -2. Charge is -2.
\( y + 4(-2) = -2 \)
\( y - 8 = -2 \)
\( y = 6 \).
Step3: Calculate change
Change = 6 - 4 = +2. So increase of 2.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a increase of 2