QUESTION IMAGE
Question
question 31 of 49
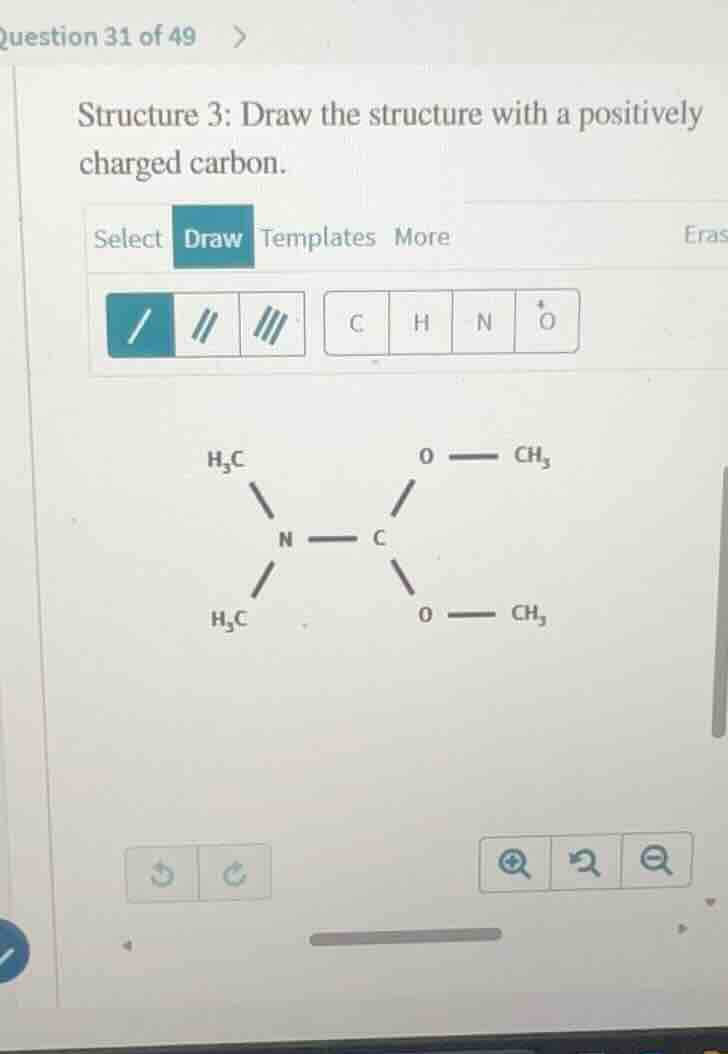
structure 3: draw the structure with a positively charged carbon.
select draw templates more eras
/ 〃 〃〃 c h n o
h₃c o — ch₃
n — c
h₃c o — ch₃
The original structure has a carbon bonded to two OCH₃ groups and a N(CH₃)₂ group, with four bonds (neutral charge). To make the carbon positively charged, we remove one of its lone pair-bearing substituents (here, we remove one of the O-CH₃ groups, leaving the carbon with only three covalent bonds). A carbon with three bonds and no lone pairs has a +1 formal charge.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The positively charged carbon structure is:
H₃C
|
H₃C-N-C⁺(O-CH₃)
|
(removed O-CH₃)Or visually, a central carbon with three bonds: one to N(CH₃)₂, one to O-CH₃, and no fourth bond, carrying a +1 charge.